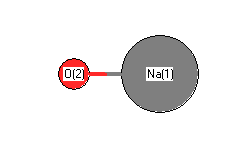
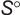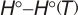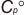.
| squib |
reference |
DOI |
| 1979HUB/HER |
Huber, K.P.; Herzberg, G., Molecular Spectra and Molecular Structure. IV. Constants of Diatomic Molecules, Van Nostrand Reinhold Co., 1979 |
10.1007/978-1-4757-0961-2 |
| 1989YAM/FUJ |
Yamada, C.; Masaharu, F.; Hirota, E., Detection of the Nao radical by microwave spectroscopy, J. Chem. Phys. 90, 3033, 1989 |
10.1063/1.455905 |
| 1989Yam/Fuj:3033-3037 |
C Yamada, M Fujitake, E Hirota "Detection of the NaO radical by microwave spectroscopy" J. Chem. Phys. 90, 3033 (1989) |
10.1063/1.455905 |
| JANAF |
Chase, M.W., Jr.; Davies, C.A.; Downey, J.R., Jr.; Frurip, D.J.; McDonald, R.A.; Syverud, A.N., JANAF Thermochemical Tables (Third Edition), J. Phys. Chem. Ref. Data,Suppl. 1, 1985, 14, 1. |
|
| webbook |
NIST Chemistry Webbook (http://webbook.nist.gov/chemistry) |
10.18434/T4D303 |