|
|
IV.A.4. (XIV.F.) |
Relative enthalpies of isomers - Comparison of 0K enthalpies (kJ mol-1)
Isomers of C3H4O2
2015 06 30 15:39| index | Species | CAS number | Name | Relative experimental enthalpy (kJ mol-1) | sketch |
|---|---|---|---|---|---|
| a | C3H4O2 | 57578 | β–Propiolactone | 28.7 | 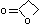 |
| b | C3H4O2 | 78988 | Methyl glyoxal | 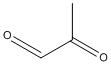 |
|
| c | C3H4O2 | 79107 | 2-Propenoic acid | 0.0 | 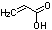 |
| d | C3H4O2 | 542789 | propanedial | 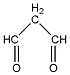 |
|
| e | C3H4O2 | 9000128 | propenalol | 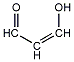 |
|
| f | HOCHCCHOH | 9000400 | allenediol | 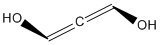 |
| composite | G1 | NC NC NC NC |
|---|---|---|
| G2MP2 | NC NC NC NC |
|
| G2 | NC NC |
|
| G3 | 43.4 a 41.5 b 0.0 c 187.5 f |
|
| G3B3 | 43.6 a 41.3 b 0.0 c |
|
| G3MP2 | 46.0 a 0.0 c |
|
| G4 | NC NC |
|
| CBS-Q | 40.9 a 39.6 b 0.0 c 168.2 f |
| STO-3G | 3-21G | 3-21G* | 6-31G | 6-31G* | 6-31G** | 6-31+G** | 6-311G* | 6-311G** | 6-31G(2df,p) | 6-311+G(3df,2p) | TZVP | cc-pVDZ | cc-pVTZ | aug-cc-pVDZ | aug-cc-pVTZ | cc-pV(T+d)Z | daug-cc-pVTZ | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| hartree fock | HF | -105.6 a -21.1 b 0.0 c 1.9 d 29.0 e 179.7 f |
61.8 a 56.9 b 0.0 c 78.5 d 51.2 e 199.0 f |
61.8 a 56.9 b 0.0 c 78.5 d 51.2 e 199.0 f |
93.7 a 52.0 b 0.0 c 70.9 d 53.3 e 198.1 f |
36.5 a 26.4 b 0.0 c 44.0 d 59.0 e 235.5 f |
50.0 a 39.4 b 0.0 c 56.9 d 58.9 e 223.2 f |
56.6 a 43.3 b 0.0 c 59.0 d 59.4 e 217.0 f |
41.7 a 26.7 b 0.0 c 42.8 d 62.0 e 233.1 f |
56.4 a 41.3 b 0.0 c 217.2 f |
45.5 a 37.2 b 0.0 c 55.4 d 60.2 e 220.1 f |
52.4 a 0.0 c 56.7 d 60.4 e |
56.1 a 41.8 b 0.0 c 59.3 e 211.8 f |
56.4 a 38.4 b 0.0 c 56.0 d 58.6 e 221.8 f |
54.4 a 43.7 b 0.0 c 59.0 d 60.7 e 214.1 f |
53.7 a 40.3 b 0.0 c 54.6 d 58.0 e 212.3 f |
53.8 a 43.6 b 0.0 c 211.1 f |
54.4 a 0.0 c 59.0 d 60.7 e |
53.7 a 43.6 b 0.0 c 58.4 d 59.9 e 211.1 f |
| density functional | LSDA | NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC |
NC NC NC NC |
NC NC |
NC NC |
|||
| BLYP | -96.4 a 0.0 c 20.8 d -69.2 e 171.7 f |
20.5 a 0.0 c 56.2 d -1.5 e 183.6 f |
20.5 a 0.0 c 56.2 d -1.5 e 183.6 f |
48.7 a 32.0 b 0.0 c 56.2 d 19.6 e 181.1 f |
21.7 a 22.3 b 0.0 c 43.1 d 21.2 e 189.3 f |
32.4 a 32.8 b 0.0 c 53.9 d 19.9 e 180.4 f |
42.9 a 38.8 b 0.0 c 56.9 d 26.6 e 172.3 f |
33.4 a 25.7 b 0.0 c 45.5 d 29.8 e 187.6 f |
46.5 a 38.7 b 0.0 c 174.7 f |
31.5 a 34.9 b 0.0 c 55.6 d 20.9 e 173.9 f |
NC NC |
38.6 a 35.6 b 0.0 c 56.9 d 18.1 e 177.7 f |
46.9 a 43.0 b 0.0 c 60.8 d 28.0 e 169.1 f |
43.9 a 0.0 c |
46.9 a 0.0 c 60.8 d 28.0 e |
||||
| B1B95 | -110.1 a -1.2 b 0.0 c 180.2 f |
15.1 a 53.2 b 0.0 c 202.3 f |
15.1 a 53.2 b 0.0 c 202.3 f |
43.2 a 0.0 c 200.8 f |
5.3 a 0.0 c 198.2 f |
17.1 a 0.0 c 236.3 f |
24.8 a 0.0 c 63.5 d 34.0 e 194.3 f |
13.1 a 0.0 c 211.3 f |
27.4 a 0.0 c 196.5 f |
14.5 a 54.2 b 0.0 c 193.8 f |
21.8 a 53.9 b 0.0 c 197.8 f |
25.0 a 0.0 c 177.3 f |
24.1 a 0.0 c 175.5 f |
NC NC |
25.0 a 0.0 c |
||||
| B3LYP | -101.3 a -7.4 b 0.0 c 17.4 d -52.8 e 173.6 f |
26.4 a 0.0 c 61.4 d 13.1 e 188.9 f |
26.4 a 0.0 c 61.4 d 13.2 e 188.9 f |
54.8 a 37.1 b 0.0 c 60.6 d 28.4 e 185.5 f |
20.5 a 25.8 b 0.0 c 47.2 d 30.3 e 198.3 f |
32.0 a 36.9 b 0.0 c 58.4 d 29.4 e 188.7 f |
41.5 a 42.4 b 0.0 c 61.6 d 34.1 e 177.1 f |
31.3 a 28.6 b 0.0 c 49.1 d 37.4 e 196.7 f |
45.1 a 42.2 b 0.0 c 182.9 f |
30.9 a 38.5 b 0.0 c 59.9 d 30.2 e 182.5 f |
44.2 a 0.0 c |
46.9 a 43.0 b 0.0 c 35.0 e 178.2 f |
38.2 a 38.9 b 0.0 c 60.0 d 28.0 e 186.3 f |
45.0 a 46.2 b 0.0 c 35.2 e 177.7 f |
42.6 a 45.1 b 0.0 c 62.9 d 32.6 e 174.9 f |
45.3 a 47.0 b 0.0 c 65.0 d 35.0 e 173.6 f |
45.0 a 0.0 c 35.2 e |
||
| B3LYPultrafine | 20.5 a 25.8 b 0.0 c 198.4 f |
NC NC |
NC NC NC |
NC NC |
45.3 a 47.1 b 0.0 c 65.0 d 35.0 e 173.7 f |
||||||||||||||
| B3PW91 | -109.8 a -7.7 b 0.0 c 16.0 d -54.2 e 176.1 f |
16.4 a 0.0 c 64.0 d 10.4 e 187.6 f |
16.4 a 0.0 c 64.0 d 10.4 e 187.6 f |
44.5 a 39.4 b 0.0 c 62.0 d 27.5 e 184.7 f |
8.0 a 28.8 b 0.0 c 49.2 d 29.5 e 196.9 f |
19.8 a 40.2 b 0.0 c 60.7 d 27.8 e 186.8 f |
27.2 a 44.3 b 0.0 c 62.7 d 31.6 e 180.3 f |
16.1 a 30.1 b 0.0 c 49.3 d 35.8 e 196.3 f |
30.7 a 44.3 b 0.0 c 181.8 f |
18.1 a 41.3 b 0.0 c 61.9 d 28.3 e 181.3 f |
NC NC |
24.5 a 40.9 b 0.0 c 60.9 d 25.7 e 185.2 f |
29.3 a 48.0 b 0.0 c 65.9 d 32.2 e 176.2 f |
26.9 a 0.0 c |
29.3 a 0.0 c 65.9 d 32.2 e |
||||
| mPW1PW91 | -112.3 a -8.2 b 0.0 c 15.9 d -50.7 e 177.0 f |
16.1 a 0.0 c 60.7 d 7.9 e 184.4 f |
16.2 a 0.0 c 65.7 d 12.8 e 189.4 f |
44.4 a 40.6 b 0.0 c 63.0 d 28.9 e 186.4 f |
6.1 a 25.0 b 0.0 c 45.4 d 26.5 e 195.1 f |
18.2 a 36.7 b 0.0 c 57.0 d 25.0 e 184.7 f |
25.7 a 41.0 b 0.0 c 59.5 d 28.9 e 178.4 f |
13.9 a 26.1 b 0.0 c 45.0 d 32.7 e 194.7 f |
28.6 a 45.0 b 0.0 c 184.4 f |
16.2 a 42.2 b 0.0 c 62.7 d 29.9 e 184.0 f |
NC NC |
22.9 a 37.0 b 0.0 c 56.9 d 22.8 e 183.3 f |
27.3 a 48.7 b 0.0 c 66.8 d 33.5 e 178.7 f |
24.9 a 46.3 b 0.0 c 177.3 f |
NC NC |
27.3 a 0.0 c 66.8 d 33.5 e |
|||
| M06-2X | NC NC |
NC NC |
28.0 a 46.5 b 0.0 c 69.2 d 21.7 e 185.9 f |
NC NC |
18.0 a 33.0 b 0.0 c 40.1 e 199.2 f |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
||||
| PBEPBE | -109.6 a 0.0 c 22.6 d -76.4 e 174.9 f |
5.2 a 0.0 c 59.7 d -11.8 e 184.0 f |
5.2 a 0.0 c 59.7 d -11.8 e 184.0 f |
34.2 a 35.5 b 0.0 c 58.9 d 15.0 e 182.2 f |
3.8 a 26.8 b 0.0 c 46.6 d 17.0 e 189.0 f |
15.2 a 37.8 b 0.0 c 57.9 d 14.2 e 179.3 f |
24.1 a 42.7 b 0.0 c 60.1 d 20.6 e 172.4 f |
12.7 a 28.7 b 0.0 c 47.2 d 25.4 e 188.3 f |
27.0 a 42.6 b 0.0 c 174.5 f |
13.3 a 39.6 b 0.0 c 59.3 d 14.7 e 173.6 f |
NC NC |
20.3 a 39.4 b 0.0 c 59.7 d 11.5 e 177.5 f |
26.2 a 47.0 b 0.0 c 63.8 d 20.9 e 168.0 f |
24.0 a 45.5 b 0.0 c 166.4 f |
NC NC |
26.2 a 0.0 c 63.8 d 20.9 e |
|||
| PBEPBEultrafine | 3.8 a 26.8 b 0.0 c 189.0 f |
NC NC |
NC NC |
NC NC |
NC NC |
||||||||||||||
| PBE1PBE | NC NC |
NC NC |
3.9 a 30.0 b 0.0 c 30.3 e 200.0 f |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
||||||
| HSEh1PBE | NC NC |
16.3 a 0.0 c 11.3 e 189.7 f |
NC NC |
6.6 a 29.9 b 0.0 c 29.9 e 199.8 f |
NC NC |
26.5 a 0.0 c 32.3 e |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
28.1 a 49.4 b 0.0 c 32.8 e 178.5 f |
NC NC |
NC NC |
|||||
| TPSSh | 0.8 a 0.0 c 44.6 d 24.7 e |
19.4 a 0.0 c 57.4 d 27.0 e |
9.6 a 35.3 b 0.0 c 55.8 d 23.2 e 184.4 f |
20.7 a 0.0 c 59.7 d 27.2 e |
|||||||||||||||
| wB97X-D | 20.5 a 37.4 b 0.0 c 61.8 d 22.6 e 196.2 f |
8.5 a 0.0 c 36.4 e 207.3 f |
28.0 a 0.0 c 38.6 e 190.4 f |
31.0 a 0.0 c 60.3 d 39.3 e 191.8 f |
31.3 a 0.0 c 57.3 d 38.3 e 187.0 f |
28.0 a 0.0 c 38.6 e 190.4 f |
29.0 a 0.0 c 62.0 d 39.0 e 187.1 f |
28.8 a 42.7 b 0.0 c 61.6 d 38.9 e 183.8 f |
|||||||||||
| B97D3 | 23.7 a 0.0 c 60.2 d 0.3 e 186.7 f |
21.2 a 25.3 b 0.0 c 44.4 d 22.2 e 193.9 f |
40.8 a 41.2 b 0.0 c 57.9 d 25.8 e 177.5 f |
43.2 a 40.5 b 0.0 c 58.9 d 26.4 e 179.4 f |
42.1 a 43.5 b 0.0 c 59.0 d 27.7 e 172.2 f |
45.1 a 41.4 b 0.0 c 56.7 d 26.1 e 174.2 f |
42.7 a 44.3 b 0.0 c 60.5 d 26.2 e 173.4 f |
43.0 a 45.1 b 0.0 c 60.2 d 26.4 e 169.6 f |
|||||||||||
| STO-3G | 3-21G | 3-21G* | 6-31G | 6-31G* | 6-31G** | 6-31+G** | 6-311G* | 6-311G** | 6-31G(2df,p) | 6-311+G(3df,2p) | TZVP | cc-pVDZ | cc-pVTZ | aug-cc-pVDZ | aug-cc-pVTZ | cc-pV(T+d)Z | daug-cc-pVTZ | ||
| Moller Plesset perturbation | MP2 | -79.3 a -52.3 b 0.0 c -28.6 d 15.5 e 217.5 f |
39.4 a 20.9 b 0.0 c 47.5 d 40.0 e 212.5 f |
39.4 a 20.9 b 0.0 c 47.5 d 40.0 e 212.5 f |
68.5 a 21.2 b 0.0 c 46.9 d 49.3 e 207.8 f |
20.4 a 22.8 b 0.0 c 44.5 d 45.2 e 220.4 f |
29.2 a 31.6 b 0.0 c 54.3 d 44.6 e 212.2 f |
NC NC NC NC |
21.8 a 20.9 b 0.0 c 42.1 d 52.2 e 222.1 f |
36.6 a 36.2 b 0.0 c 202.6 f |
23.6 a 38.5 b 0.0 c 61.0 d 44.3 e 203.4 f |
40.0 a 37.9 b 0.0 c 46.1 e 199.7 f |
36.5 a 32.9 b 0.0 c 54.8 d 42.6 e 208.6 f |
NC NC NC NC NC |
NC NC NC NC |
NC NC NC |
NC NC |
||
| MP2=FULL | NC NC NC NC |
NC NC NC NC |
NC NC NC NC |
NC NC NC NC |
20.0 a 23.3 b 0.0 c 45.2 d 45.3 e 220.4 f |
28.6 a 31.9 b 0.0 c 54.8 d 44.7 e 212.2 f |
37.1 a 37.4 b 0.0 c 58.4 d 46.3 e 203.6 f |
21.6 a 21.5 b 0.0 c 44.0 d 52.2 e 221.7 f |
36.1 a 36.7 b 0.0 c 202.2 f |
NC NC NC NC |
NC NC |
NC NC NC NC |
NC NC NC NC NC |
NC NC |
NC NC |
NC NC |
|||
| MP3 | 28.0 a 28.5 b 0.0 c 211.0 f |
||||||||||||||||||
| MP3=FULL | 27.5 a 0.0 c 49.3 d 51.8 e |
||||||||||||||||||
| MP4 | NC NC NC |
NC NC NC NC |
NC NC |
NC NC |
NC NC |
||||||||||||||
| MP4=FULL | NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
||||||||||||||
| B2PLYP | 21.1 a 0.0 c 36.1 e |
42.1 a 0.0 c 64.6 d 38.4 e |
|||||||||||||||||
| Configuration interaction | CID | NC NC NC NC |
NC NC NC NC |
NC NC NC NC |
24.2 a 27.5 b 0.0 c 46.5 d 53.3 e 222.6 f |
NC NC NC NC |
|||||||||||||
| CISD | NC NC NC NC |
NC NC NC NC |
NC NC NC NC |
23.7 a 27.0 b 0.0 c 46.0 d 52.9 e 223.7 f |
NC NC NC NC |
||||||||||||||
| STO-3G | 3-21G | 3-21G* | 6-31G | 6-31G* | 6-31G** | 6-31+G** | 6-311G* | 6-311G** | 6-31G(2df,p) | 6-311+G(3df,2p) | TZVP | cc-pVDZ | cc-pVTZ | aug-cc-pVDZ | aug-cc-pVTZ | cc-pV(T+d)Z | daug-cc-pVTZ | ||
| Quadratic configuration interaction | QCISD | NC NC |
NC NC NC NC NC |
NC NC NC NC |
NC NC NC NC |
NC NC NC NC NC |
NC NC NC NC NC |
NC NC NC NC NC |
NC NC NC NC |
NC NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
||||
| QCISD(T) | NC NC NC NC |
NC NC |
NC NC |
NC NC |
|||||||||||||||
| Coupled Cluster | CCD | NC NC |
NC NC NC NC |
NC NC NC NC |
NC NC NC NC |
NC NC NC NC NC |
NC NC NC NC |
NC NC NC NC |
NC NC NC NC |
NC NC |
NC NC NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
NC NC |
|||
| CCSD | NC NC NC NC |
NC NC |
NC NC |
||||||||||||||||
| CCSD=FULL | NC NC |
NC NC |
NC NC |
||||||||||||||||
| CCSD(T) | NC NC NC NC |
NC NC |
NC NC |
||||||||||||||||
| CCSD(T)=FULL | NC NC |
NC NC |
NC NC |
||||||||||||||||
| STO-3G | 3-21G | 3-21G* | 6-31G | 6-31G* | 6-31G** | 6-31+G** | 6-311G* | 6-311G** | 6-31G(2df,p) | 6-311+G(3df,2p) | TZVP | cc-pVDZ | cc-pVTZ | aug-cc-pVDZ | aug-cc-pVTZ | cc-pV(T+d)Z | daug-cc-pVTZ |
| CEP-31G | CEP-31G* | CEP-121G | CEP-121G* | LANL2DZ | SDD | cc-pVTZ-PP | aug-cc-pVTZ-PP | Def2TZVPP | ||
|---|---|---|---|---|---|---|---|---|---|---|
| hartree fock | HF | 72.9 a 40.8 b 0.0 c 57.0 d 48.4 e 209.3 f |
20.0 a 18.8 b 0.0 c 33.7 d 59.4 e 251.8 f |
78.4 a 45.5 b 0.0 c 60.8 d 48.1 e 187.1 f |
35.0 a 26.2 b 0.0 c 41.5 d 59.9 e 227.5 f |
85.1 a 50.4 b 0.0 c 68.6 d 49.8 e 186.6 f |
84.7 a 50.4 b 0.0 c 68.6 d 50.0 e 186.6 f |
54.9 a 44.0 b 0.0 c 58.9 d 60.4 e 212.3 f |
||
| density functional | B1B95 | 22.7 a 0.0 c 58.8 d 26.2 e |
-12.5 a 0.0 c 47.4 d 32.7 e |
|||||||
| B3LYP | 38.9 a 32.9 b 0.0 c 55.0 d 27.7 e 190.0 f |
9.0 a 22.6 b 0.0 c 42.6 d 33.5 e 210.1 f |
47.5 a 36.1 b 0.0 c 56.9 d 25.9 e 173.1 f |
23.6 a 27.9 b 0.0 c 47.3 d 33.9 e 191.3 f |
47.7 a 38.9 b 0.0 c 63.2 d 25.5 e 171.5 f |
47.7 a 38.8 b 0.0 c 63.0 d 25.9 e 172.3 f |
45.5 a 46.6 b 0.0 c 64.9 d 35.7 e 176.1 f |
|||
| PBEPBE | 26.7 a 47.4 b 0.0 c 63.3 d 21.7 e 166.5 f |
|||||||||
| Moller Plesset perturbation | MP2 | 54.0 a 16.0 b 0.0 c 40.5 d 48.4 e 212.2 f |
4.8 a 16.1 b 0.0 c 37.9 d 50.1 e 235.7 f |
62.0 a 21.3 b 0.0 c 43.3 d 46.2 e 196.6 f |
20.4 a 24.0 b 0.0 c 44.0 d 48.5 e 214.5 f |
65.6 a 25.0 b 0.0 c 50.4 d 47.0 e 192.3 f |
65.3 a 24.9 b 0.0 c 50.3 d 47.3 e 192.5 f |
34.5 a 47.0 b 0.0 c 66.0 d 43.3 e 191.4 f |
| 6-311+G(3df,2p) | cc-pVDZ | cc-pVTZ | aug-cc-pVDZ | aug-cc-pVTZ | cc-pV(T+d)Z | ||
|---|---|---|---|---|---|---|---|
| Moller Plesset perturbation | MP2FC// HF/6-31G* | 35.6 a 0.0 c |
39.8 a 0.0 c |
39.8 a 0.0 c |
|||
| MP2FC// B3LYP/6-31G* | 36.1 a 0.0 c |
||||||
| MP2FC// MP2FC/6-31G* | 34.5 a 0.0 c |
37.9 a 0.0 c |
35.9 a 0.0 c |
40.1 a 0.0 c |
36.0 a 0.0 c 67.4 d 44.1 e |
35.9 a 0.0 c |
|
| MP4// HF/6-31G* | 37.7 a 0.0 c |
||||||
| MP4// MP2/6-31G* | 35.3 a 0.0 c |
||||||
| Coupled Cluster | CCSD// HF/6-31G* | 40.4 a 0.0 c |
|||||
| CCSD(T)// HF/6-31G* | 39.6 a 0.0 c |
||||||
| CCSD(T)//B3LYP/6-31G(2df,p) | 38.1 a 0.0 c 64.2 d 45.6 e |
38.1 a 0.0 c 64.2 d 45.6 e |
|||||
| CCSD// MP2FC/6-31G* | 38.0 a 0.0 c |
40.9 a 0.0 c |
40.1 a 0.0 c 63.2 d 51.2 e |
40.9 a 0.0 c |
|||
| CCSD(T)// MP2FC/6-31G* | 38.0 a 0.0 c |
40.4 a 0.0 c |
40.2 a 0.0 c 65.4 d 46.7 e |
40.4 a 0.0 c |
For descriptions of the methods (AM1, HF, MP2, ...) and basis sets (3-21G, 3-21G*, 6-31G, ...) see the glossary in section I.C. Predefined means the basis set used is determined by the method.
gaw refers to the group additivity method implemeted in the NIST Chemistry Webbook.
See section III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.