|
|
II. |
Experimental thermochemical data
| A. | Single molecule | ||
| 1. | All experimental data for one molecule | ||
| 2. | Types of data available for one molecule | ||
| 3. | Summary of geometry data for one molecule | ||
| B. | Specific experimental properties | ||
| 1. | Atomization energies | ||
| 2. | Vibrational frequencies  | ||
| a. | Anharmonic constants for Diatomics  | ||
| b. | Anharmonic constants for Triatomics  | ||
| 3. | Bond lengths  | ||
| 4. | Bond angles  | ||
| 5. | Rotational constants  | ||
| 6. | Experimental barriers to internal rotation  | ||
| C. | Reference data | ||
| 1. | Thermodynamic reference states | ||
| 2. | List of electronic spin splitting corrections | ||
| 3. | List of species with experimental enthalpy of formation at 0K | ||
| 4. | Suggestions for species with well-defined experimental enthalpies of formation | ||
| D. | One experimental property for several species | ||
| 1. | Enthalpy of formation  | ||
| 2. | Entropy at 298.15K 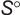 | ||
| 3. | Integrated Heat Capacity 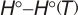 | ||
| 4. | Heat Capacity at 298.15K 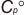 | ||
| 5. | Rotational Constants  | ||
| 6. | Vibrational Frequencies  | ||
| 7. | Electric dipole moment  | ||
| 8. | Ionization Energy  | ||
| 9. | Polarizability  | ||
| 10. | Quadrupole  | ||
| 11. | Point Group | ||