All results from a given calculation for C4H9SH (1-Butanethiol)
using model chemistry: CISD/cc-pVTZ
19 10 17 12 22
States and conformations
| State |
Conformation |
minimum conformation |
conformer description |
state description |
| 1 |
1 |
yes |
CS |
1A' |
Energy calculated at CISD/cc-pVTZ
| | hartrees |
|---|
| Energy at 0K | -555.667609 |
| Energy at 298.15K | |
| HF Energy | -554.900729 |
| Nuclear repulsion energy | 224.900344 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at CISD/cc-pVTZ
Geometric Data calculated at CISD/cc-pVTZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| S1 |
1.377 |
-1.834 |
0.000 |
| C2 |
-0.227 |
-0.981 |
0.000 |
| C3 |
0.000 |
0.518 |
0.000 |
| C4 |
-1.305 |
1.297 |
0.000 |
| C5 |
-1.083 |
2.799 |
0.000 |
| H6 |
0.913 |
-3.078 |
0.000 |
| H7 |
-0.785 |
-1.275 |
0.878 |
| H8 |
-0.785 |
-1.275 |
-0.878 |
| H9 |
0.587 |
0.793 |
-0.870 |
| H10 |
0.587 |
0.793 |
0.870 |
| H11 |
-1.891 |
1.016 |
0.869 |
| H12 |
-1.891 |
1.016 |
-0.869 |
| H13 |
-2.023 |
3.336 |
0.000 |
| H14 |
-0.524 |
3.109 |
0.875 |
| H15 |
-0.524 |
3.109 |
-0.875 |
Atom - Atom Distances (Å)
| |
S1 |
C2 |
C3 |
C4 |
C5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
H12 |
H13 |
H14 |
H15 |
| S1 | | 1.8161 | 2.7252 | 4.1222 | 5.2455 | 1.3282 | 2.3993 | 2.3993 | 2.8775 | 2.8775 | 4.4214 | 4.4214 | 6.1875 | 5.3672 | 5.3672 |
C2 | 1.8161 | | 1.5165 | 2.5205 | 3.8763 | 2.3867 | 1.0808 | 1.0808 | 2.1372 | 2.1372 | 2.7405 | 2.7405 | 4.6761 | 4.1932 | 4.1932 | C3 | 2.7252 | 1.5165 | | 1.5196 | 2.5252 | 3.7105 | 2.1452 | 2.1452 | 1.0851 | 1.0851 | 2.1394 | 2.1394 | 3.4689 | 2.7842 | 2.7842 | C4 | 4.1222 | 2.5205 | 1.5196 | | 1.5183 | 4.9052 | 2.7668 | 2.7668 | 2.1425 | 2.1425 | 1.0853 | 1.0853 | 2.1618 | 2.1581 | 2.1581 | C5 | 5.2455 | 3.8763 | 2.5252 | 1.5183 | | 6.2072 | 4.1782 | 4.1782 | 2.7518 | 2.7518 | 2.1421 | 2.1421 | 1.0823 | 1.0835 | 1.0835 | H6 | 1.3282 | 2.3867 | 3.7105 | 4.9052 | 6.2072 | | 2.6279 | 2.6279 | 3.9812 | 3.9812 | 5.0372 | 5.0372 | 7.0543 | 6.4118 | 6.4118 | H7 | 2.3993 | 1.0808 | 2.1452 | 2.7668 | 4.1782 | 2.6279 | | 1.7551 | 3.0355 | 2.4818 | 2.5432 | 3.0852 | 4.8542 | 4.3916 | 4.7283 | H8 | 2.3993 | 1.0808 | 2.1452 | 2.7668 | 4.1782 | 2.6279 | 1.7551 | | 2.4818 | 3.0355 | 3.0852 | 2.5432 | 4.8542 | 4.7283 | 4.3916 | H9 | 2.8775 | 2.1372 | 1.0851 | 2.1425 | 2.7518 | 3.9812 | 3.0355 | 2.4818 | | 1.7405 | 3.0354 | 2.4876 | 3.7467 | 3.1054 | 2.5687 | H10 | 2.8775 | 2.1372 | 1.0851 | 2.1425 | 2.7518 | 3.9812 | 2.4818 | 3.0355 | 1.7405 | | 2.4876 | 3.0354 | 3.7467 | 2.5687 | 3.1054 | H11 | 4.4214 | 2.7405 | 2.1394 | 1.0853 | 2.1421 | 5.0372 | 2.5432 | 3.0852 | 3.0354 | 2.4876 | | 1.7382 | 2.4816 | 2.5000 | 3.0481 | H12 | 4.4214 | 2.7405 | 2.1394 | 1.0853 | 2.1421 | 5.0372 | 3.0852 | 2.5432 | 2.4876 | 3.0354 | 1.7382 | | 2.4816 | 3.0481 | 2.5000 | H13 | 6.1875 | 4.6761 | 3.4689 | 2.1618 | 1.0823 | 7.0543 | 4.8542 | 4.8542 | 3.7467 | 3.7467 | 2.4816 | 2.4816 | | 1.7504 | 1.7504 | H14 | 5.3672 | 4.1932 | 2.7842 | 2.1581 | 1.0835 | 6.4118 | 4.3916 | 4.7283 | 3.1054 | 2.5687 | 2.5000 | 3.0481 | 1.7504 | | 1.7495 | H15 | 5.3672 | 4.1932 | 2.7842 | 2.1581 | 1.0835 | 6.4118 | 4.7283 | 4.3916 | 2.5687 | 3.1054 | 3.0481 | 2.5000 | 1.7504 | 1.7495 | |
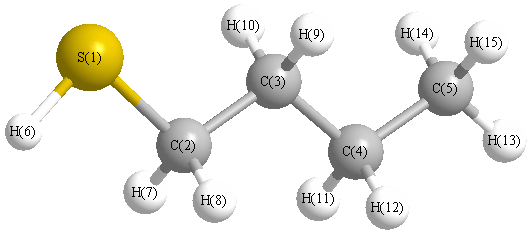 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| S1 |
C2 |
C3 |
109.386 |
|
S1 |
C2 |
H7 |
109.191 |
| S1 |
C2 |
H8 |
109.191 |
|
C2 |
S1 |
H6 |
97.548 |
| C2 |
C3 |
C4 |
112.231 |
|
C2 |
C3 |
H9 |
109.349 |
| C2 |
C3 |
H10 |
109.349 |
|
C3 |
C2 |
H7 |
110.237 |
| C3 |
C2 |
H8 |
110.237 |
|
C3 |
C4 |
C5 |
112.451 |
| C3 |
C4 |
H11 |
109.299 |
|
C3 |
C4 |
H12 |
109.299 |
| C4 |
C3 |
H9 |
109.554 |
|
C4 |
C3 |
H10 |
109.554 |
| C4 |
C5 |
H13 |
111.356 |
|
C4 |
C5 |
H14 |
110.981 |
| C4 |
C5 |
H15 |
110.981 |
|
C5 |
C4 |
H11 |
109.597 |
| C5 |
C4 |
H12 |
109.597 |
|
H7 |
C2 |
H8 |
108.576 |
| H9 |
C3 |
H10 |
106.645 |
|
H11 |
C4 |
H12 |
106.414 |
| H13 |
C5 |
H14 |
107.843 |
|
H13 |
C5 |
H15 |
107.843 |
| H14 |
C5 |
H15 |
107.678 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
