Jump to
S1C2
Energy calculated at MP2=FULL/cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -193.764005 |
| Energy at 298.15K | -193.772990 |
| Nuclear repulsion energy | 130.206078 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2=FULL/cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3859 |
3668 |
29.63 |
|
|
|
| 2 |
A' |
3188 |
3030 |
24.29 |
|
|
|
| 3 |
A' |
3102 |
2949 |
21.89 |
|
|
|
| 4 |
A' |
3085 |
2932 |
26.67 |
|
|
|
| 5 |
A' |
3023 |
2873 |
60.58 |
|
|
|
| 6 |
A' |
1531 |
1455 |
3.70 |
|
|
|
| 7 |
A' |
1511 |
1436 |
6.03 |
|
|
|
| 8 |
A' |
1494 |
1420 |
0.40 |
|
|
|
| 9 |
A' |
1476 |
1403 |
3.17 |
|
|
|
| 10 |
A' |
1416 |
1345 |
0.34 |
|
|
|
| 11 |
A' |
1349 |
1283 |
9.87 |
|
|
|
| 12 |
A' |
1276 |
1213 |
51.36 |
|
|
|
| 13 |
A' |
1124 |
1068 |
27.63 |
|
|
|
| 14 |
A' |
1097 |
1042 |
43.76 |
|
|
|
| 15 |
A' |
1078 |
1025 |
23.03 |
|
|
|
| 16 |
A' |
906 |
861 |
4.32 |
|
|
|
| 17 |
A' |
463 |
440 |
10.17 |
|
|
|
| 18 |
A' |
272 |
259 |
3.92 |
|
|
|
| 19 |
A" |
3182 |
3024 |
49.97 |
|
|
|
| 20 |
A" |
3158 |
3001 |
5.79 |
|
|
|
| 21 |
A" |
3069 |
2917 |
59.83 |
|
|
|
| 22 |
A" |
1501 |
1427 |
6.89 |
|
|
|
| 23 |
A" |
1325 |
1259 |
0.11 |
|
|
|
| 24 |
A" |
1273 |
1209 |
0.09 |
|
|
|
| 25 |
A" |
1194 |
1135 |
1.58 |
|
|
|
| 26 |
A" |
904 |
859 |
2.96 |
|
|
|
| 27 |
A" |
775 |
737 |
0.54 |
|
|
|
| 28 |
A" |
288 |
273 |
105.79 |
|
|
|
| 29 |
A" |
247 |
235 |
6.22 |
|
|
|
| 30 |
A" |
133 |
127 |
4.96 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 24148.3 cm
-1
Scaled (by 0.9504) Zero Point Vibrational Energy (zpe) 22950.5 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2=FULL/cc-pVDZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-1.448 |
1.227 |
0.000 |
| C2 |
0.000 |
0.739 |
0.000 |
| C3 |
0.099 |
-0.777 |
0.000 |
| O4 |
1.478 |
-1.123 |
0.000 |
| H5 |
-1.499 |
2.327 |
0.000 |
| H6 |
-1.990 |
0.867 |
0.891 |
| H7 |
-1.990 |
0.867 |
-0.891 |
| H8 |
0.536 |
1.120 |
0.886 |
| H9 |
0.536 |
1.120 |
-0.886 |
| H10 |
-0.422 |
-1.180 |
0.893 |
| H11 |
-0.422 |
-1.180 |
-0.893 |
| H12 |
1.523 |
-2.088 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
O4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
H12 |
| C1 | | 1.5283 | 2.5323 | 3.7532 | 1.1010 | 1.1028 | 1.1028 | 2.1758 | 2.1758 | 2.7651 | 2.7651 | 4.4516 |
C2 | 1.5283 | | 1.5196 | 2.3774 | 2.1836 | 2.1837 | 2.1837 | 1.1032 | 1.1032 | 2.1582 | 2.1582 | 3.2109 | C3 | 2.5323 | 1.5196 | | 1.4215 | 3.4915 | 2.8037 | 2.8037 | 2.1389 | 2.1389 | 1.1093 | 1.1093 | 1.9350 | O4 | 3.7532 | 2.3774 | 1.4215 | | 4.5569 | 4.0962 | 4.0962 | 2.5888 | 2.5888 | 2.0997 | 2.0997 | 0.9656 | H5 | 1.1010 | 2.1836 | 3.4915 | 4.5569 | | 1.7792 | 1.7792 | 2.5266 | 2.5266 | 3.7759 | 3.7759 | 5.3499 | H6 | 1.1028 | 2.1837 | 2.8037 | 4.0962 | 1.7792 | | 1.7812 | 2.5387 | 3.0984 | 2.5787 | 3.1353 | 4.6758 | H7 | 1.1028 | 2.1837 | 2.8037 | 4.0962 | 1.7792 | 1.7812 | | 3.0984 | 2.5387 | 3.1353 | 2.5787 | 4.6758 | H8 | 2.1758 | 1.1032 | 2.1389 | 2.5888 | 2.5266 | 2.5387 | 3.0984 | | 1.7712 | 2.4915 | 3.0611 | 3.4708 | H9 | 2.1758 | 1.1032 | 2.1389 | 2.5888 | 2.5266 | 3.0984 | 2.5387 | 1.7712 | | 3.0611 | 2.4915 | 3.4708 | H10 | 2.7651 | 2.1582 | 1.1093 | 2.0997 | 3.7759 | 2.5787 | 3.1353 | 2.4915 | 3.0611 | | 1.7857 | 2.3244 | H11 | 2.7651 | 2.1582 | 1.1093 | 2.0997 | 3.7759 | 3.1353 | 2.5787 | 3.0611 | 2.4915 | 1.7857 | | 2.3244 | H12 | 4.4516 | 3.2109 | 1.9350 | 0.9656 | 5.3499 | 4.6758 | 4.6758 | 3.4708 | 3.4708 | 2.3244 | 2.3244 | |
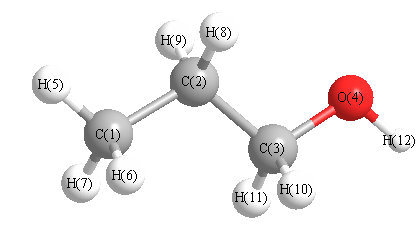 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
112.372 |
|
C1 |
C2 |
H8 |
110.515 |
| C1 |
C2 |
H9 |
110.515 |
|
C2 |
C1 |
H5 |
111.266 |
| C2 |
C1 |
H6 |
111.163 |
|
C2 |
C1 |
H7 |
111.163 |
| C2 |
C3 |
O4 |
107.824 |
|
C2 |
C3 |
H10 |
109.366 |
| C2 |
C3 |
H11 |
109.366 |
|
C3 |
C2 |
H8 |
108.225 |
| C3 |
C2 |
H9 |
108.225 |
|
C3 |
O4 |
H12 |
106.759 |
| O4 |
C3 |
H10 |
111.534 |
|
O4 |
C3 |
H11 |
111.534 |
| H5 |
C1 |
H6 |
107.678 |
|
H5 |
C1 |
H7 |
107.678 |
| H6 |
C1 |
H7 |
107.719 |
|
H8 |
C2 |
H9 |
106.788 |
| H10 |
C3 |
H11 |
107.194 |
|
Electronic energy levels
Electronic state
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at MP2=FULL/cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -193.764964 |
| Energy at 298.15K | |
| HF Energy | -193.128976 |
| Nuclear repulsion energy | 132.721666 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2=FULL/cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3862 |
3670 |
33.48 |
99.72 |
0.30 |
0.46 |
| 2 |
A |
3210 |
3051 |
16.32 |
43.48 |
0.75 |
0.86 |
| 3 |
A |
3183 |
3025 |
35.97 |
56.13 |
0.65 |
0.79 |
| 4 |
A |
3151 |
2995 |
19.04 |
88.55 |
0.74 |
0.85 |
| 5 |
A |
3097 |
2943 |
40.99 |
142.53 |
0.04 |
0.08 |
| 6 |
A |
3090 |
2937 |
38.01 |
84.84 |
0.05 |
0.10 |
| 7 |
A |
3074 |
2922 |
42.47 |
91.37 |
0.74 |
0.85 |
| 8 |
A |
3027 |
2876 |
64.44 |
108.68 |
0.11 |
0.20 |
| 9 |
A |
1527 |
1451 |
2.81 |
5.84 |
0.69 |
0.82 |
| 10 |
A |
1512 |
1437 |
7.28 |
7.36 |
0.75 |
0.85 |
| 11 |
A |
1495 |
1421 |
7.13 |
16.83 |
0.75 |
0.86 |
| 12 |
A |
1477 |
1403 |
3.73 |
13.75 |
0.71 |
0.83 |
| 13 |
A |
1470 |
1397 |
1.91 |
1.57 |
0.27 |
0.42 |
| 14 |
A |
1416 |
1345 |
3.59 |
1.72 |
0.72 |
0.84 |
| 15 |
A |
1377 |
1309 |
1.07 |
1.23 |
0.75 |
0.86 |
| 16 |
A |
1333 |
1267 |
22.93 |
11.21 |
0.74 |
0.85 |
| 17 |
A |
1279 |
1215 |
1.72 |
7.49 |
0.74 |
0.85 |
| 18 |
A |
1261 |
1198 |
37.07 |
3.32 |
0.60 |
0.75 |
| 19 |
A |
1173 |
1115 |
7.01 |
0.82 |
0.19 |
0.32 |
| 20 |
A |
1147 |
1090 |
20.73 |
3.87 |
0.56 |
0.72 |
| 21 |
A |
1101 |
1046 |
25.35 |
3.27 |
0.71 |
0.83 |
| 22 |
A |
1005 |
955 |
39.31 |
3.66 |
0.74 |
0.85 |
| 23 |
A |
933 |
886 |
2.32 |
0.36 |
0.38 |
0.55 |
| 24 |
A |
897 |
853 |
1.85 |
9.83 |
0.15 |
0.26 |
| 25 |
A |
780 |
742 |
0.59 |
0.38 |
0.54 |
0.70 |
| 26 |
A |
482 |
459 |
7.59 |
0.23 |
0.46 |
0.63 |
| 27 |
A |
336 |
319 |
10.90 |
0.42 |
0.59 |
0.75 |
| 28 |
A |
270 |
257 |
97.10 |
3.82 |
0.74 |
0.85 |
| 29 |
A |
244 |
232 |
4.61 |
0.10 |
0.72 |
0.84 |
| 30 |
A |
150 |
143 |
6.96 |
0.13 |
0.75 |
0.86 |
Unscaled Zero Point Vibrational Energy (zpe) 24178.6 cm
-1
Scaled (by 0.9504) Zero Point Vibrational Energy (zpe) 22979.3 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2=FULL/cc-pVDZ
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-1.521 |
-0.520 |
0.130 |
| C2 |
-0.632 |
0.652 |
-0.288 |
| C3 |
0.771 |
0.548 |
0.289 |
| O4 |
1.356 |
-0.645 |
-0.220 |
| H5 |
-2.523 |
-0.445 |
-0.321 |
| H6 |
-1.644 |
-0.546 |
1.226 |
| H7 |
-1.066 |
-1.471 |
-0.182 |
| H8 |
-0.549 |
0.690 |
-1.387 |
| H9 |
-1.076 |
1.609 |
0.038 |
| H10 |
0.712 |
0.527 |
1.397 |
| H11 |
1.362 |
1.440 |
0.004 |
| H12 |
2.224 |
-0.729 |
0.195 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
O4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
H12 |
| C1 | | 1.5284 | 2.5331 | 2.9010 | 1.1012 | 1.1031 | 1.0996 | 2.1705 | 2.1770 | 2.7726 | 3.4883 | 3.7513 |
C2 | 1.5284 | | 1.5210 | 2.3751 | 2.1857 | 2.1789 | 2.1690 | 1.1037 | 1.1040 | 2.1593 | 2.1644 | 3.2092 | C3 | 2.5331 | 1.5210 | | 1.4226 | 3.4937 | 2.8113 | 2.7697 | 2.1388 | 2.1449 | 1.1095 | 1.1080 | 1.9364 | O4 | 2.9010 | 2.3751 | 1.4226 | | 3.8854 | 3.3318 | 2.5596 | 2.6029 | 3.3259 | 2.0981 | 2.0970 | 0.9655 | H5 | 1.1012 | 2.1857 | 3.4937 | 3.8854 | | 1.7820 | 1.7869 | 2.5143 | 2.5384 | 3.7897 | 4.3303 | 4.7831 | H6 | 1.1031 | 2.1789 | 2.8113 | 3.3318 | 1.7820 | | 1.7813 | 3.0913 | 2.5258 | 2.5943 | 3.8042 | 4.0070 | H7 | 1.0996 | 2.1690 | 2.7697 | 2.5596 | 1.7869 | 1.7813 | | 2.5278 | 3.0880 | 3.1060 | 3.7954 | 3.3937 | H8 | 2.1705 | 1.1037 | 2.1388 | 2.6029 | 2.5143 | 3.0913 | 2.5278 | | 1.7756 | 3.0613 | 2.4801 | 3.4939 | H9 | 2.1770 | 1.1040 | 2.1449 | 3.3259 | 2.5384 | 2.5258 | 3.0880 | 1.7756 | | 2.4935 | 2.4438 | 4.0472 | H10 | 2.7726 | 2.1593 | 1.1095 | 2.0981 | 3.7897 | 2.5943 | 3.1060 | 3.0613 | 2.4935 | | 1.7882 | 2.3037 | H11 | 3.4883 | 2.1644 | 1.1080 | 2.0970 | 4.3303 | 3.8042 | 3.7954 | 2.4801 | 2.4438 | 1.7882 | | 2.3419 | H12 | 3.7513 | 3.2092 | 1.9364 | 0.9655 | 4.7831 | 4.0070 | 3.3937 | 3.4939 | 4.0472 | 2.3037 | 2.3419 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
112.343 |
|
C1 |
C2 |
H8 |
110.052 |
| C1 |
C2 |
H9 |
110.550 |
|
C2 |
C1 |
H5 |
111.409 |
| C2 |
C1 |
H6 |
110.751 |
|
C2 |
C1 |
H7 |
110.184 |
| C2 |
C3 |
O4 |
107.534 |
|
C2 |
C3 |
H10 |
109.346 |
| C2 |
C3 |
H11 |
109.838 |
|
C3 |
C2 |
H8 |
108.095 |
| C3 |
C2 |
H9 |
108.550 |
|
C3 |
O4 |
H12 |
106.793 |
| O4 |
C3 |
H10 |
111.310 |
|
O4 |
C3 |
H11 |
111.314 |
| H5 |
C1 |
H6 |
107.883 |
|
H5 |
C1 |
H7 |
108.571 |
| H6 |
C1 |
H7 |
107.933 |
|
H8 |
C2 |
H9 |
107.077 |
| H10 |
C3 |
H11 |
107.491 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
