Jump to
S1C2
Energy calculated at MP2/TZVP
| | hartrees |
|---|
| Energy at 0K | -1495.936623 |
| Energy at 298.15K | |
| HF Energy | -1495.069109 |
| Nuclear repulsion energy | 425.785372 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2/TZVP
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3225 |
3057 |
2.31 |
|
|
|
| 2 |
A' |
3175 |
3009 |
2.88 |
|
|
|
| 3 |
A' |
3144 |
2980 |
13.70 |
|
|
|
| 4 |
A' |
1505 |
1427 |
4.94 |
|
|
|
| 5 |
A' |
1397 |
1324 |
22.17 |
|
|
|
| 6 |
A' |
1311 |
1243 |
1.39 |
|
|
|
| 7 |
A' |
1262 |
1197 |
25.80 |
|
|
|
| 8 |
A' |
1082 |
1026 |
1.57 |
|
|
|
| 9 |
A' |
900 |
853 |
10.16 |
|
|
|
| 10 |
A' |
853 |
808 |
0.75 |
|
|
|
| 11 |
A' |
758 |
719 |
86.98 |
|
|
|
| 12 |
A' |
401 |
380 |
0.32 |
|
|
|
| 13 |
A' |
349 |
331 |
0.22 |
|
|
|
| 14 |
A' |
174 |
165 |
6.17 |
|
|
|
| 15 |
A' |
124 |
117 |
2.03 |
|
|
|
| 16 |
A" |
3222 |
3054 |
0.81 |
|
|
|
| 17 |
A" |
3144 |
2980 |
0.11 |
|
|
|
| 18 |
A" |
1485 |
1408 |
6.84 |
|
|
|
| 19 |
A" |
1419 |
1345 |
0.02 |
|
|
|
| 20 |
A" |
1342 |
1272 |
1.37 |
|
|
|
| 21 |
A" |
1197 |
1134 |
0.98 |
|
|
|
| 22 |
A" |
1157 |
1097 |
0.00 |
|
|
|
| 23 |
A" |
889 |
843 |
2.98 |
|
|
|
| 24 |
A" |
788 |
747 |
10.33 |
|
|
|
| 25 |
A" |
257 |
244 |
10.32 |
|
|
|
| 26 |
A" |
254 |
241 |
0.86 |
|
|
|
| 27 |
A" |
34i |
32i |
1.37 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 17389.4 cm
-1
Scaled (by 0.9479) Zero Point Vibrational Energy (zpe) 16483.4 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2/TZVP
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.453 |
0.693 |
0.000 |
| C2 |
-0.184 |
0.219 |
1.329 |
| C3 |
-0.184 |
0.219 |
-1.329 |
| Cl4 |
0.353 |
2.448 |
0.000 |
| Cl5 |
-0.184 |
-1.508 |
-1.637 |
| Cl6 |
-0.184 |
-1.508 |
1.637 |
| H7 |
1.494 |
0.377 |
0.000 |
| H8 |
0.349 |
0.683 |
-2.148 |
| H9 |
0.349 |
0.683 |
2.148 |
| H10 |
-1.230 |
0.561 |
-1.348 |
| H11 |
-1.230 |
0.561 |
1.348 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
Cl4 |
Cl5 |
Cl6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5480 | 1.5480 | 1.7578 | 2.8161 | 2.8161 | 1.0880 | 2.1505 | 2.1505 | 2.1605 | 2.1605 |
C2 | 1.5480 | | 2.6580 | 2.6500 | 3.4321 | 1.7548 | 2.1461 | 3.5480 | 1.0814 | 2.8946 | 1.1008 | C3 | 1.5480 | 2.6580 | | 2.6500 | 1.7548 | 3.4321 | 2.1461 | 1.0814 | 3.5480 | 1.1008 | 2.8946 | Cl4 | 1.7578 | 2.6500 | 2.6500 | | 4.3152 | 4.3152 | 2.3647 | 2.7801 | 2.7801 | 2.8083 | 2.8083 | Cl5 | 2.8161 | 3.4321 | 1.7548 | 4.3152 | | 3.2732 | 3.0079 | 2.3125 | 4.4056 | 2.3364 | 3.7795 | Cl6 | 2.8161 | 1.7548 | 3.4321 | 4.3152 | 3.2732 | | 3.0079 | 4.4056 | 2.3125 | 3.7795 | 2.3364 | H7 | 1.0880 | 2.1461 | 2.1461 | 2.3647 | 3.0079 | 3.0079 | | 2.4534 | 2.4534 | 3.0450 | 3.0450 | H8 | 2.1505 | 3.5480 | 1.0814 | 2.7801 | 2.3125 | 4.4056 | 2.4534 | | 4.2960 | 1.7741 | 3.8381 | H9 | 2.1505 | 1.0814 | 3.5480 | 2.7801 | 4.4056 | 2.3125 | 2.4534 | 4.2960 | | 3.8381 | 1.7741 | H10 | 2.1605 | 2.8946 | 1.1008 | 2.8083 | 2.3364 | 3.7795 | 3.0450 | 1.7741 | 3.8381 | | 2.6964 | H11 | 2.1605 | 1.1008 | 2.8946 | 2.8083 | 3.7795 | 2.3364 | 3.0450 | 3.8381 | 1.7741 | 2.6964 | |
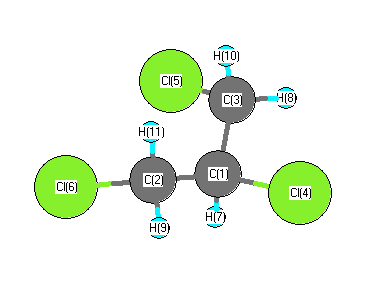 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
Cl6 |
116.868 |
|
C1 |
C2 |
H9 |
108.440 |
| C1 |
C2 |
H11 |
108.120 |
|
C1 |
C3 |
Cl5 |
116.868 |
| C1 |
C3 |
H8 |
108.440 |
|
C1 |
C3 |
H10 |
108.120 |
| C2 |
C1 |
C3 |
118.306 |
|
C2 |
C1 |
Cl4 |
106.397 |
| C2 |
C1 |
H7 |
107.731 |
|
C3 |
C1 |
Cl4 |
106.397 |
| C3 |
C1 |
H7 |
107.731 |
|
Cl4 |
C1 |
H7 |
110.157 |
| Cl5 |
C3 |
H8 |
106.832 |
|
Cl5 |
C3 |
H10 |
107.593 |
| Cl6 |
C2 |
H9 |
106.832 |
|
Cl6 |
C2 |
H11 |
107.593 |
| H8 |
C3 |
H10 |
108.772 |
|
H9 |
C2 |
H11 |
108.772 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at MP2/TZVP
| | hartrees |
|---|
| Energy at 0K | -1495.936722 |
| Energy at 298.15K | |
| HF Energy | -1495.069038 |
| Nuclear repulsion energy | 426.151298 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2/TZVP
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3227 |
3058 |
1.59 |
|
|
|
| 2 |
A |
3222 |
3054 |
1.53 |
|
|
|
| 3 |
A |
3177 |
3011 |
2.25 |
|
|
|
| 4 |
A |
3145 |
2981 |
1.29 |
|
|
|
| 5 |
A |
3144 |
2980 |
12.61 |
|
|
|
| 6 |
A |
1505 |
1427 |
5.04 |
|
|
|
| 7 |
A |
1488 |
1410 |
7.30 |
|
|
|
| 8 |
A |
1414 |
1340 |
0.23 |
|
|
|
| 9 |
A |
1392 |
1320 |
19.61 |
|
|
|
| 10 |
A |
1338 |
1268 |
3.14 |
|
|
|
| 11 |
A |
1302 |
1235 |
0.51 |
|
|
|
| 12 |
A |
1267 |
1201 |
27.25 |
|
|
|
| 13 |
A |
1194 |
1132 |
0.90 |
|
|
|
| 14 |
A |
1153 |
1093 |
0.80 |
|
|
|
| 15 |
A |
1078 |
1022 |
2.04 |
|
|
|
| 16 |
A |
908 |
860 |
2.79 |
|
|
|
| 17 |
A |
899 |
852 |
8.70 |
|
|
|
| 18 |
A |
841 |
797 |
1.50 |
|
|
|
| 19 |
A |
789 |
748 |
10.92 |
|
|
|
| 20 |
A |
758 |
719 |
84.91 |
|
|
|
| 21 |
A |
405 |
384 |
0.96 |
|
|
|
| 22 |
A |
348 |
330 |
0.26 |
|
|
|
| 23 |
A |
262 |
248 |
10.19 |
|
|
|
| 24 |
A |
255 |
241 |
0.35 |
|
|
|
| 25 |
A |
170 |
161 |
5.44 |
|
|
|
| 26 |
A |
115 |
109 |
2.33 |
|
|
|
| 27 |
A |
49 |
46 |
2.18 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 17421.1 cm
-1
Scaled (by 0.9479) Zero Point Vibrational Energy (zpe) 16513.5 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2/TZVP
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.688 |
-0.019 |
-0.291 |
| C2 |
0.111 |
-1.203 |
0.462 |
| C3 |
0.269 |
1.344 |
0.212 |
| Cl4 |
2.488 |
-0.075 |
-0.074 |
| Cl5 |
-1.483 |
1.646 |
-0.030 |
| Cl6 |
-1.533 |
-1.636 |
-0.114 |
| H7 |
0.490 |
-0.115 |
-1.355 |
| H8 |
0.806 |
2.116 |
-0.330 |
| H9 |
0.735 |
-2.075 |
0.302 |
| H10 |
0.475 |
1.428 |
1.276 |
| H11 |
0.048 |
-0.984 |
1.525 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
Cl4 |
Cl5 |
Cl6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5162 | 1.5122 | 1.8146 | 2.7480 | 2.7518 | 1.0867 | 2.1385 | 2.1402 | 2.1436 | 2.1533 |
C2 | 1.5162 | | 2.5638 | 2.6848 | 3.3010 | 1.7948 | 2.1510 | 3.4813 | 1.0842 | 2.7775 | 1.0874 | C3 | 1.5122 | 2.5638 | | 2.6493 | 1.7946 | 3.4975 | 2.1532 | 1.0848 | 3.4519 | 1.0870 | 2.6817 | Cl4 | 1.8146 | 2.6848 | 2.6493 | | 4.3282 | 4.3132 | 2.3736 | 2.7738 | 2.6864 | 2.8520 | 3.0558 | Cl5 | 2.7480 | 3.3010 | 1.7946 | 4.3282 | | 3.2827 | 2.9585 | 2.3559 | 4.3443 | 2.3638 | 3.4169 | Cl6 | 2.7518 | 1.7948 | 3.4975 | 4.3132 | 3.2827 | | 2.8185 | 4.4257 | 2.3468 | 3.9176 | 2.3684 | H7 | 1.0867 | 2.1510 | 2.1532 | 2.3736 | 2.9585 | 2.8185 | | 2.4756 | 2.5781 | 3.0506 | 3.0406 | H8 | 2.1385 | 3.4813 | 1.0848 | 2.7738 | 2.3559 | 4.4257 | 2.4756 | | 4.2387 | 1.7783 | 3.6906 | H9 | 2.1402 | 1.0842 | 3.4519 | 2.6864 | 4.3443 | 2.3468 | 2.5781 | 4.2387 | | 3.6450 | 1.7772 | H10 | 2.1436 | 2.7775 | 1.0870 | 2.8520 | 2.3638 | 3.9176 | 3.0506 | 1.7783 | 3.6450 | | 2.4615 | H11 | 2.1533 | 1.0874 | 2.6817 | 3.0558 | 3.4169 | 2.3684 | 3.0406 | 3.6906 | 1.7772 | 2.4615 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
Cl6 |
112.154 |
|
C1 |
C2 |
H9 |
109.663 |
| C1 |
C2 |
H11 |
110.513 |
|
C1 |
C3 |
Cl5 |
112.124 |
| C1 |
C3 |
H8 |
109.769 |
|
C1 |
C3 |
H10 |
110.041 |
| C2 |
C1 |
C3 |
115.688 |
|
C2 |
C1 |
Cl4 |
107.088 |
| C2 |
C1 |
H7 |
110.376 |
|
C3 |
C1 |
Cl4 |
105.204 |
| C3 |
C1 |
H7 |
110.828 |
|
Cl4 |
C1 |
H7 |
107.114 |
| Cl5 |
C3 |
H8 |
107.220 |
|
Cl5 |
C3 |
H10 |
107.690 |
| Cl6 |
C2 |
H9 |
106.580 |
|
Cl6 |
C2 |
H11 |
107.986 |
| H8 |
C3 |
H10 |
109.930 |
|
H9 |
C2 |
H11 |
109.847 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
