Jump to
S1C2
Energy calculated at PBE1PBE/6-311+G(3df,2p)
| | hartrees |
|---|
| Energy at 0K | -188.966492 |
| Energy at 298.15K | -188.967595 |
| HF Energy | -188.966492 |
| Nuclear repulsion energy | 63.615838 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at PBE1PBE/6-311+G(3df,2p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3662 |
3662 |
23.09 |
|
|
|
| 2 |
A' |
1915 |
1915 |
356.86 |
|
|
|
| 3 |
A' |
1304 |
1304 |
0.15 |
|
|
|
| 4 |
A' |
1110 |
1110 |
164.68 |
|
|
|
| 5 |
A' |
606 |
606 |
34.37 |
|
|
|
| 6 |
A" |
599 |
599 |
108.31 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 4597.8 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 4597.8 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at PBE1PBE/6-311+G(3df,2p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.000 |
0.435 |
0.000 |
| O2 |
-1.055 |
-0.350 |
0.000 |
| O3 |
1.151 |
0.184 |
0.000 |
| H4 |
-0.767 |
-1.279 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
O3 |
H4 |
| C1 | | 1.3147 | 1.1776 | 1.8779 |
O2 | 1.3147 | | 2.2689 | 0.9727 | O3 | 1.1776 | 2.2689 | | 2.4121 | H4 | 1.8779 | 0.9727 | 2.4121 | |
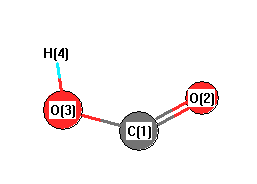 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
O2 |
H4 |
109.461 |
|
O2 |
C1 |
O3 |
131.038 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at PBE1PBE/6-311+G(3df,2p)
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
0.677 |
|
|
|
| 2 |
O |
-0.389 |
|
|
|
| 3 |
O |
-0.508 |
|
|
|
| 4 |
H |
0.219 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
-1.110 |
-1.675 |
0.000 |
2.009 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-20.214 |
0.797 |
0.000 |
| y |
0.797 |
-14.074 |
0.000 |
| z |
0.000 |
0.000 |
-16.370 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
-4.992 |
0.797 |
0.000 |
| y |
0.797 |
4.218 |
0.000 |
| z |
0.000 |
0.000 |
0.774 |
|
| Polar |
|---|
| 3z2-r2 | 1.548 |
|---|
| x2-y2 | -6.140 |
|---|
| xy | 0.797 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
3.890 |
0.182 |
0.000 |
| y |
0.182 |
3.396 |
0.000 |
| z |
0.000 |
0.000 |
2.344 |
<r2> (average value of r
2) Å
2
| <r2> |
34.645 |
| (<r2>)1/2 |
5.886 |
Jump to
S1C1
Energy calculated at PBE1PBE/6-311+G(3df,2p)
| | hartrees |
|---|
| Energy at 0K | -188.969206 |
| Energy at 298.15K | -188.970295 |
| HF Energy | -188.969206 |
| Nuclear repulsion energy | 63.380067 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at PBE1PBE/6-311+G(3df,2p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3870 |
3870 |
134.82 |
|
|
|
| 2 |
A' |
1950 |
1950 |
265.73 |
|
|
|
| 3 |
A' |
1253 |
1253 |
269.11 |
|
|
|
| 4 |
A' |
1127 |
1127 |
44.40 |
|
|
|
| 5 |
A' |
630 |
630 |
4.34 |
|
|
|
| 6 |
A" |
556 |
556 |
85.46 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 4692.9 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 4692.9 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at PBE1PBE/6-311+G(3df,2p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.000 |
0.403 |
0.000 |
| O2 |
-0.938 |
-0.539 |
0.000 |
| O3 |
1.163 |
0.251 |
0.000 |
| H4 |
-1.801 |
-0.115 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
O3 |
H4 |
| C1 | | 1.3288 | 1.1727 | 1.8740 |
O2 | 1.3288 | | 2.2442 | 0.9618 | O3 | 1.1727 | 2.2442 | | 2.9865 | H4 | 1.8740 | 0.9618 | 2.9865 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
O2 |
H4 |
108.738 |
|
O2 |
C1 |
O3 |
127.465 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at PBE1PBE/6-311+G(3df,2p)
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
0.631 |
|
|
|
| 2 |
O |
-0.392 |
|
|
|
| 3 |
O |
-0.489 |
|
|
|
| 4 |
H |
0.250 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
-3.029 |
0.295 |
0.000 |
3.044 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-14.071 |
-0.940 |
0.000 |
| y |
-0.940 |
-18.116 |
0.000 |
| z |
0.000 |
0.000 |
-16.423 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
3.199 |
-0.940 |
0.000 |
| y |
-0.940 |
-2.869 |
0.000 |
| z |
0.000 |
0.000 |
-0.330 |
|
| Polar |
|---|
| 3z2-r2 | -0.659 |
|---|
| x2-y2 | 4.045 |
|---|
| xy | -0.940 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
4.269 |
0.123 |
0.000 |
| y |
0.123 |
2.973 |
0.000 |
| z |
0.000 |
0.000 |
2.398 |
<r2> (average value of r
2) Å
2
| <r2> |
35.030 |
| (<r2>)1/2 |
5.919 |
