Jump to
S1C2
Energy calculated at HSEh1PBE/6-31G(2df,p)
| | hartrees |
|---|
| Energy at 0K | -114.302159 |
| Energy at 298.15K | -114.303577 |
| HF Energy | -114.302159 |
| Nuclear repulsion energy | 30.793098 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at HSEh1PBE/6-31G(2df,p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3753 |
3595 |
60.20 |
|
|
|
| 2 |
A' |
2831 |
2712 |
144.60 |
|
|
|
| 3 |
A' |
1522 |
1458 |
20.43 |
|
|
|
| 4 |
A' |
1369 |
1311 |
63.34 |
|
|
|
| 5 |
A' |
1223 |
1171 |
125.21 |
|
|
|
| 6 |
A" |
1106 |
1059 |
107.39 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 5902.5 cm
-1
Scaled (by 0.9577) Zero Point Vibrational Energy (zpe) 5652.8 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at HSEh1PBE/6-31G(2df,p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.011 |
0.740 |
0.000 |
| O2 |
0.011 |
-0.567 |
0.000 |
| H3 |
-1.089 |
0.965 |
0.000 |
| H4 |
0.930 |
-0.867 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
H3 |
H4 |
| C1 | | 1.3070 | 1.1235 | 1.8512 |
O2 | 1.3070 | | 1.8869 | 0.9665 | H3 | 1.1235 | 1.8869 | | 2.7270 | H4 | 1.8512 | 0.9665 | 2.7270 | |
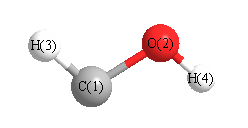 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
O2 |
H4 |
108.086 |
|
O2 |
C1 |
H3 |
101.585 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at HSEh1PBE/6-31G(2df,p)
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
-0.223 |
|
|
|
| 2 |
O |
-0.244 |
|
|
|
| 3 |
H |
0.124 |
|
|
|
| 4 |
H |
0.342 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
0.324 |
-1.668 |
0.000 |
1.699 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-10.761 |
-3.374 |
0.000 |
| y |
-3.374 |
-13.385 |
0.000 |
| z |
0.000 |
0.000 |
-11.694 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
1.778 |
-3.374 |
0.000 |
| y |
-3.374 |
-2.157 |
0.000 |
| z |
0.000 |
0.000 |
0.378 |
|
| Polar |
|---|
| 3z2-r2 | 0.757 |
|---|
| x2-y2 | 2.624 |
|---|
| xy | -3.374 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
2.527 |
-0.565 |
0.000 |
| y |
-0.565 |
2.506 |
0.000 |
| z |
0.000 |
0.000 |
1.611 |
<r2> (average value of r
2) Å
2
| <r2> |
17.057 |
| (<r2>)1/2 |
4.130 |
Jump to
S1C1
Energy calculated at HSEh1PBE/6-31G(2df,p)
| | hartrees |
|---|
| Energy at 0K | -114.295248 |
| Energy at 298.15K | -114.296656 |
| HF Energy | -114.295248 |
| Nuclear repulsion energy | 30.747510 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at HSEh1PBE/6-31G(2df,p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3604 |
3452 |
17.27 |
|
|
|
| 2 |
A' |
2757 |
2641 |
203.14 |
|
|
|
| 3 |
A' |
1486 |
1423 |
47.59 |
|
|
|
| 4 |
A' |
1379 |
1321 |
60.58 |
|
|
|
| 5 |
A' |
1230 |
1178 |
28.72 |
|
|
|
| 6 |
A" |
1031 |
988 |
32.72 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 5744.0 cm
-1
Scaled (by 0.9577) Zero Point Vibrational Energy (zpe) 5501.0 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at HSEh1PBE/6-31G(2df,p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.122 |
0.738 |
0.000 |
| O2 |
0.122 |
-0.563 |
0.000 |
| H3 |
-0.958 |
1.064 |
0.000 |
| H4 |
-0.753 |
-0.988 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
H3 |
H4 |
| C1 | | 1.3015 | 1.1280 | 1.9353 |
O2 | 1.3015 | | 1.9531 | 0.9727 | H3 | 1.1280 | 1.9531 | | 2.0621 | H4 | 1.9353 | 0.9727 | 2.0621 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
O2 |
H4 |
115.881 |
|
O2 |
C1 |
H3 |
106.789 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at HSEh1PBE/6-31G(2df,p)
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
-0.180 |
|
|
|
| 2 |
O |
-0.243 |
|
|
|
| 3 |
H |
0.099 |
|
|
|
| 4 |
H |
0.325 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
-2.751 |
-1.727 |
0.000 |
3.249 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-11.937 |
-0.036 |
0.000 |
| y |
-0.036 |
-12.856 |
0.000 |
| z |
0.000 |
0.000 |
-11.680 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
0.331 |
-0.036 |
0.000 |
| y |
-0.036 |
-1.047 |
0.000 |
| z |
0.000 |
0.000 |
0.716 |
|
| Polar |
|---|
| 3z2-r2 | 1.433 |
|---|
| x2-y2 | 0.919 |
|---|
| xy | -0.036 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
2.399 |
-0.073 |
0.000 |
| y |
-0.073 |
2.901 |
0.000 |
| z |
0.000 |
0.000 |
1.625 |
<r2> (average value of r
2) Å
2
| <r2> |
17.203 |
| (<r2>)1/2 |
4.148 |
