Jump to
S1C2
Vibrational Frequencies calculated at B2PLYP=FULL/TZVP
Geometric Data calculated at B2PLYP=FULL/TZVP
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at B2PLYP=FULL/TZVP
| | hartrees |
|---|
| Energy at 0K | -998.707997 |
| Energy at 298.15K | -998.712844 |
| HF Energy | -998.490841 |
| Nuclear repulsion energy | 201.050422 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at B2PLYP=FULL/TZVP
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3176 |
3030 |
0.83 |
|
|
|
| 2 |
A |
3117 |
2974 |
23.54 |
|
|
|
| 3 |
A |
1489 |
1420 |
0.64 |
|
|
|
| 4 |
A |
1372 |
1309 |
22.49 |
|
|
|
| 5 |
A |
1246 |
1189 |
1.22 |
|
|
|
| 6 |
A |
1066 |
1017 |
0.93 |
|
|
|
| 7 |
A |
970 |
925 |
10.26 |
|
|
|
| 8 |
A |
667 |
636 |
21.06 |
|
|
|
| 9 |
A |
266 |
254 |
0.77 |
|
|
|
| 10 |
A |
117 |
112 |
0.96 |
|
|
|
| 11 |
B |
3188 |
3042 |
5.11 |
|
|
|
| 12 |
B |
3109 |
2967 |
3.41 |
|
|
|
| 13 |
B |
1484 |
1416 |
11.14 |
|
|
|
| 14 |
B |
1349 |
1288 |
46.42 |
|
|
|
| 15 |
B |
1179 |
1125 |
1.22 |
|
|
|
| 16 |
B |
910 |
869 |
17.27 |
|
|
|
| 17 |
B |
691 |
660 |
26.56 |
|
|
|
| 18 |
B |
414 |
395 |
8.75 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 12904.1 cm
-1
Scaled (by 0.9542) Zero Point Vibrational Energy (zpe) 12313.1 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at B2PLYP=FULL/TZVP
Point Group is C2
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.298 |
0.692 |
0.897 |
| C2 |
-0.298 |
-0.692 |
0.897 |
| Cl3 |
-0.298 |
1.706 |
-0.472 |
| Cl4 |
0.298 |
-1.706 |
-0.472 |
| H5 |
0.009 |
1.205 |
1.812 |
| H6 |
1.380 |
0.658 |
0.824 |
| H7 |
-0.009 |
-1.205 |
1.812 |
| H8 |
-1.380 |
-0.658 |
0.824 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
Cl3 |
Cl4 |
H5 |
H6 |
H7 |
H8 |
| C1 | | 1.5070 | 1.8041 | 2.7611 | 1.0876 | 1.0852 | 2.1281 | 2.1549 |
C2 | 1.5070 | | 2.7611 | 1.8041 | 2.1281 | 2.1549 | 1.0876 | 1.0852 | Cl3 | 1.8041 | 2.7611 | | 3.4632 | 2.3578 | 2.3639 | 3.7105 | 2.9049 | Cl4 | 2.7611 | 1.8041 | 3.4632 | | 3.7105 | 2.9049 | 2.3578 | 2.3639 | H5 | 1.0876 | 2.1281 | 2.3578 | 3.7105 | | 1.7761 | 2.4092 | 2.5251 | H6 | 1.0852 | 2.1549 | 2.3639 | 2.9049 | 1.7761 | | 2.5251 | 3.0577 | H7 | 2.1281 | 1.0876 | 3.7105 | 2.3578 | 2.4092 | 2.5251 | | 1.7761 | H8 | 2.1549 | 1.0852 | 2.9049 | 2.3639 | 2.5251 | 3.0577 | 1.7761 | |
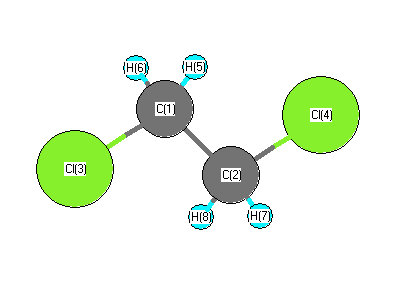 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
Cl4 |
112.696 |
|
C1 |
C2 |
H7 |
109.142 |
| C1 |
C2 |
H8 |
111.428 |
|
C2 |
C1 |
Cl3 |
112.696 |
| C2 |
C1 |
H5 |
109.142 |
|
C2 |
C1 |
H6 |
111.428 |
| Cl3 |
C1 |
H5 |
106.607 |
|
Cl3 |
C1 |
H6 |
107.164 |
| Cl4 |
C2 |
H7 |
106.607 |
|
Cl4 |
C2 |
H8 |
107.164 |
| H5 |
C1 |
H6 |
109.657 |
|
H7 |
C2 |
H8 |
109.657 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
