Jump to
S1C2
Energy calculated at CCD/6-31G**
| | hartrees |
|---|
| Energy at 0K | -134.773522 |
| Energy at 298.15K | -134.781785 |
| HF Energy | -134.262190 |
| Nuclear repulsion energy | 82.863911 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at CCD/6-31G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3563 |
3340 |
1.31 |
|
|
|
| 2 |
A' |
3193 |
2994 |
40.70 |
|
|
|
| 3 |
A' |
3123 |
2928 |
35.44 |
|
|
|
| 4 |
A' |
3105 |
2911 |
20.85 |
|
|
|
| 5 |
A' |
1717 |
1609 |
21.61 |
|
|
|
| 6 |
A' |
1568 |
1471 |
0.95 |
|
|
|
| 7 |
A' |
1551 |
1454 |
0.05 |
|
|
|
| 8 |
A' |
1474 |
1382 |
6.67 |
|
|
|
| 9 |
A' |
1433 |
1344 |
15.16 |
|
|
|
| 10 |
A' |
1198 |
1124 |
11.20 |
|
|
|
| 11 |
A' |
1116 |
1047 |
25.97 |
|
|
|
| 12 |
A' |
944 |
885 |
106.11 |
|
|
|
| 13 |
A' |
908 |
851 |
61.16 |
|
|
|
| 14 |
A' |
412 |
386 |
6.96 |
|
|
|
| 15 |
A" |
3654 |
3426 |
0.52 |
|
|
|
| 16 |
A" |
3194 |
2995 |
61.86 |
|
|
|
| 17 |
A" |
3165 |
2967 |
6.93 |
|
|
|
| 18 |
A" |
1558 |
1461 |
5.14 |
|
|
|
| 19 |
A" |
1432 |
1343 |
0.00 |
|
|
|
| 20 |
A" |
1320 |
1237 |
0.11 |
|
|
|
| 21 |
A" |
1036 |
971 |
1.54 |
|
|
|
| 22 |
A" |
797 |
747 |
0.24 |
|
|
|
| 23 |
A" |
318 |
298 |
42.72 |
|
|
|
| 24 |
A" |
270 |
253 |
13.52 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21022.6 cm
-1
Scaled (by 0.9376) Zero Point Vibrational Energy (zpe) 19710.8 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at CCD/6-31G**
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| N1 |
-1.307 |
-0.086 |
0.000 |
| C2 |
0.000 |
0.577 |
0.000 |
| C3 |
1.210 |
-0.356 |
0.000 |
| H4 |
2.146 |
0.205 |
0.000 |
| H5 |
1.202 |
-0.999 |
0.883 |
| H6 |
1.202 |
-0.999 |
-0.883 |
| H7 |
0.039 |
1.230 |
-0.874 |
| H8 |
0.039 |
1.230 |
0.874 |
| H9 |
-1.371 |
-0.696 |
0.808 |
| H10 |
-1.371 |
-0.696 |
-0.808 |
Atom - Atom Distances (Å)
| |
N1 |
C2 |
C3 |
H4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
| N1 | | 1.4653 | 2.5318 | 3.4651 | 2.8125 | 2.8125 | 2.0751 | 2.0751 | 1.0147 | 1.0147 |
C2 | 1.4653 | | 1.5283 | 2.1778 | 2.1695 | 2.1695 | 1.0919 | 1.0919 | 2.0374 | 2.0374 | C3 | 2.5318 | 1.5283 | | 1.0911 | 1.0915 | 1.0915 | 2.1570 | 2.1570 | 2.7257 | 2.7257 | H4 | 3.4651 | 2.1778 | 1.0911 | | 1.7657 | 1.7657 | 2.5008 | 2.5008 | 3.7189 | 3.7189 | H5 | 2.8125 | 2.1695 | 1.0915 | 1.7657 | | 1.7651 | 3.0670 | 2.5141 | 2.5919 | 3.0938 | H6 | 2.8125 | 2.1695 | 1.0915 | 1.7657 | 1.7651 | | 2.5141 | 3.0670 | 3.0938 | 2.5919 | H7 | 2.0751 | 1.0919 | 2.1570 | 2.5008 | 3.0670 | 2.5141 | | 1.7484 | 2.9197 | 2.3870 | H8 | 2.0751 | 1.0919 | 2.1570 | 2.5008 | 2.5141 | 3.0670 | 1.7484 | | 2.3870 | 2.9197 | H9 | 1.0147 | 2.0374 | 2.7257 | 3.7189 | 2.5919 | 3.0938 | 2.9197 | 2.3870 | | 1.6168 | H10 | 1.0147 | 2.0374 | 2.7257 | 3.7189 | 3.0938 | 2.5919 | 2.3870 | 2.9197 | 1.6168 | |
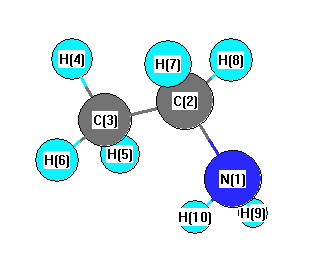 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| N1 |
C2 |
C3 |
115.492 |
|
N1 |
C2 |
H7 |
107.583 |
| N1 |
C2 |
H8 |
107.583 |
|
C2 |
N1 |
H9 |
109.124 |
| C2 |
N1 |
H10 |
109.124 |
|
C2 |
C3 |
H4 |
111.399 |
| C2 |
C3 |
H5 |
110.709 |
|
C2 |
C3 |
H6 |
110.709 |
| C3 |
C2 |
H7 |
109.698 |
|
C3 |
C2 |
H8 |
109.698 |
| H4 |
C3 |
H5 |
107.993 |
|
H4 |
C3 |
H6 |
107.993 |
| H5 |
C3 |
H6 |
107.905 |
|
H7 |
C2 |
H8 |
106.379 |
| H9 |
N1 |
H10 |
105.631 |
|
Electronic energy levels
Electronic state
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at CCD/6-31G**
| | hartrees |
|---|
| Energy at 0K | -134.773284 |
| Energy at 298.15K | |
| Nuclear repulsion energy | |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at CCD/6-31G**
Geometric Data calculated at CCD/6-31G**
Point Group is Cs
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
