All results from a given calculation for C5H9N (1,2,3,6-Tetrahydropyridine)
using model chemistry: MP3/6-31+G**
19 10 17 12 22
States and conformations
| State |
Conformation |
minimum conformation |
conformer description |
state description |
| 1 |
1 |
yes |
C1 |
1A |
Energy calculated at MP3/6-31+G**
| | hartrees |
|---|
| Energy at 0K | -249.924099 |
| Energy at 298.15K | |
| HF Energy | -249.013784 |
| Nuclear repulsion energy | 239.967302 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/6-31+G**
Geometric Data calculated at MP3/6-31+G**
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| H1 |
-1.443 |
2.073 |
0.135 |
| C2 |
-0.797 |
1.206 |
0.057 |
| H3 |
0.962 |
2.347 |
-0.093 |
| C4 |
0.528 |
1.356 |
-0.050 |
| H5 |
2.337 |
0.358 |
0.523 |
| H6 |
1.858 |
0.068 |
-1.133 |
| C7 |
1.470 |
0.178 |
-0.115 |
| H8 |
0.628 |
-1.105 |
1.401 |
| H9 |
1.351 |
-1.985 |
0.065 |
| C10 |
0.753 |
-1.108 |
0.316 |
| H11 |
-2.319 |
-0.168 |
-0.576 |
| H12 |
-1.851 |
-0.342 |
1.098 |
| C13 |
-1.457 |
-0.155 |
0.093 |
| H14 |
-0.478 |
-1.278 |
-1.285 |
| N15 |
-0.574 |
-1.260 |
-0.277 |
Atom - Atom Distances (Å)
| |
H1 |
C2 |
H3 |
C4 |
H5 |
H6 |
C7 |
H8 |
H9 |
C10 |
H11 |
H12 |
C13 |
H14 |
N15 |
| H1 | | 1.0837 | 2.4311 | 2.1050 | 4.1690 | 4.0648 | 3.4836 | 3.9981 | 4.9264 | 3.8694 | 2.5091 | 2.6313 | 2.2282 | 3.7647 | 3.4688 |
C2 | 1.0837 | | 2.1023 | 1.3376 | 3.2806 | 3.1242 | 2.4950 | 3.0292 | 3.8460 | 2.7973 | 2.1457 | 2.1426 | 1.5130 | 2.8409 | 2.4985 | H3 | 2.4311 | 2.1023 | | 1.0832 | 2.4959 | 2.6606 | 2.2283 | 3.7762 | 4.3520 | 3.4859 | 4.1624 | 4.0698 | 3.4855 | 4.0789 | 3.9253 | C4 | 2.1050 | 1.3376 | 1.0832 | | 2.1445 | 2.1451 | 1.5098 | 2.8581 | 3.4421 | 2.5013 | 3.2716 | 3.1399 | 2.4987 | 3.0782 | 2.8479 | H5 | 4.1690 | 3.2806 | 2.4959 | 2.1445 | | 1.7482 | 1.0925 | 2.4151 | 2.5826 | 2.1687 | 4.8131 | 4.2853 | 3.8531 | 3.7250 | 3.4260 | H6 | 4.0648 | 3.1242 | 2.6606 | 2.1451 | 1.7482 | | 1.0946 | 3.0513 | 2.4300 | 2.1690 | 4.2207 | 4.3479 | 3.5420 | 2.7010 | 2.9007 | C7 | 3.4836 | 2.4950 | 2.2283 | 1.5098 | 1.0925 | 1.0946 | | 2.1571 | 2.1729 | 1.5340 | 3.8320 | 3.5734 | 2.9530 | 2.6985 | 2.5043 | H8 | 3.9981 | 3.0292 | 3.7762 | 2.8581 | 2.4151 | 3.0513 | 2.1571 | | 1.7557 | 1.0924 | 3.6698 | 2.6112 | 2.6379 | 2.9103 | 2.0703 | H9 | 4.9264 | 3.8460 | 4.3520 | 3.4421 | 2.5826 | 2.4300 | 2.1729 | 1.7557 | | 1.0901 | 4.1443 | 3.7440 | 3.3515 | 2.3809 | 2.0852 | C10 | 3.8694 | 2.7973 | 3.4859 | 2.5013 | 2.1687 | 2.1690 | 1.5340 | 1.0924 | 1.0901 | | 3.3338 | 2.8249 | 2.4171 | 2.0270 | 1.4617 | H11 | 2.5091 | 2.1457 | 4.1624 | 3.2716 | 4.8131 | 4.2207 | 3.8320 | 3.6698 | 4.1443 | 3.3338 | | 1.7466 | 1.0912 | 2.2629 | 2.0796 | H12 | 2.6313 | 2.1426 | 4.0698 | 3.1399 | 4.2853 | 4.3479 | 3.5734 | 2.6112 | 3.7440 | 2.8249 | 1.7466 | | 1.0950 | 2.9050 | 2.0892 | C13 | 2.2282 | 1.5130 | 3.4855 | 2.4987 | 3.8531 | 3.5420 | 2.9530 | 2.6379 | 3.3515 | 2.4171 | 1.0912 | 1.0950 | | 2.0295 | 1.4622 | H14 | 3.7647 | 2.8409 | 4.0789 | 3.0782 | 3.7250 | 2.7010 | 2.6985 | 2.9103 | 2.3809 | 2.0270 | 2.2629 | 2.9050 | 2.0295 | | 1.0126 | N15 | 3.4688 | 2.4985 | 3.9253 | 2.8479 | 3.4260 | 2.9007 | 2.5043 | 2.0703 | 2.0852 | 1.4617 | 2.0796 | 2.0892 | 1.4622 | 1.0126 | |
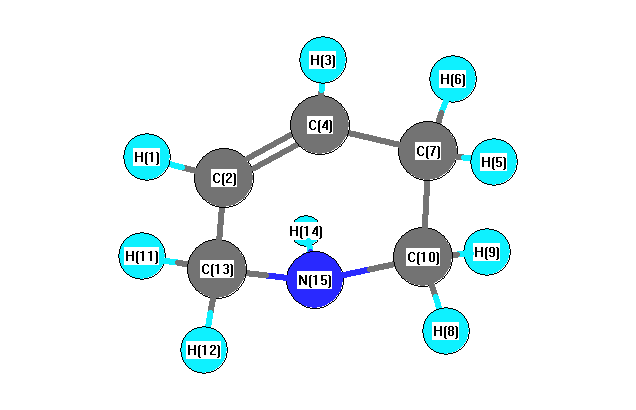 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| H1 |
C2 |
C4 |
120.408 |
|
H1 |
C2 |
C13 |
117.248 |
| C2 |
C4 |
H3 |
120.190 |
|
C2 |
C4 |
C7 |
122.264 |
| C2 |
C13 |
H11 |
109.910 |
|
C2 |
C13 |
H12 |
109.436 |
| C2 |
C13 |
N15 |
114.224 |
|
H3 |
C4 |
C7 |
117.546 |
| C4 |
C2 |
C13 |
122.338 |
|
C4 |
C7 |
H5 |
109.957 |
| C4 |
C7 |
H6 |
109.881 |
|
C4 |
C7 |
C10 |
110.517 |
| H5 |
C7 |
H6 |
106.132 |
|
H5 |
C7 |
C10 |
110.181 |
| H6 |
C7 |
C10 |
110.082 |
|
C7 |
C10 |
H8 |
109.275 |
| C7 |
C10 |
H9 |
110.655 |
|
C7 |
C10 |
N15 |
113.405 |
| H8 |
C10 |
H9 |
107.117 |
|
H8 |
C10 |
N15 |
107.424 |
| H9 |
C10 |
N15 |
108.726 |
|
C10 |
N15 |
C13 |
111.515 |
| C10 |
N15 |
H14 |
108.655 |
|
H11 |
C13 |
H12 |
106.050 |
| H11 |
C13 |
N15 |
108.180 |
|
H12 |
C13 |
N15 |
108.713 |
| C13 |
N15 |
H14 |
108.820 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
