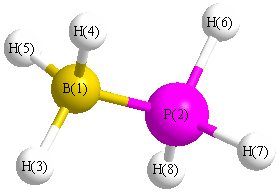All results from a given calculation for BH3PH3 (borane phosphine)
using model chemistry: CCSD=FULL/aug-cc-pVTZ
19 10 17 12 22
States and conformations
| State |
Conformation |
minimum conformation |
conformer description |
state description |
| 1 |
1 |
yes |
C3V |
1A1 |
Energy calculated at CCSD=FULL/aug-cc-pVTZ
| | hartrees |
|---|
| Energy at 0K | -369.328986 |
| Energy at 298.15K | |
| HF Energy | -368.905751 |
| Nuclear repulsion energy | 59.403566 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at CCSD=FULL/aug-cc-pVTZ
Geometric Data calculated at CCSD=FULL/aug-cc-pVTZ
Point Group is C3v
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| B1 |
0.000 |
0.000 |
-1.381 |
| P2 |
0.000 |
0.000 |
0.552 |
| H3 |
0.000 |
-1.168 |
-1.663 |
| H4 |
-1.012 |
0.584 |
-1.663 |
| H5 |
1.012 |
0.584 |
-1.663 |
| H6 |
0.000 |
1.239 |
1.202 |
| H7 |
-1.073 |
-0.620 |
1.202 |
| H8 |
1.073 |
-0.620 |
1.202 |
Atom - Atom Distances (Å)
| |
B1 |
P2 |
H3 |
H4 |
H5 |
H6 |
H7 |
H8 |
| B1 | | 1.9330 | 1.2019 | 1.2019 | 1.2019 | 2.8647 | 2.8647 | 2.8647 |
P2 | 1.9330 | | 2.5048 | 2.5048 | 2.5048 | 1.3992 | 1.3992 | 1.3992 | H3 | 1.2019 | 2.5048 | | 2.0234 | 2.0234 | 3.7425 | 3.1086 | 3.1086 | H4 | 1.2019 | 2.5048 | 2.0234 | | 2.0234 | 3.1086 | 3.1086 | 3.7425 | H5 | 1.2019 | 2.5048 | 2.0234 | 2.0234 | | 3.1086 | 3.7425 | 3.1086 | H6 | 2.8647 | 1.3992 | 3.7425 | 3.1086 | 3.1086 | | 2.1462 | 2.1462 | H7 | 2.8647 | 1.3992 | 3.1086 | 3.1086 | 3.7425 | 2.1462 | | 2.1462 | H8 | 2.8647 | 1.3992 | 3.1086 | 3.7425 | 3.1086 | 2.1462 | 2.1462 | |
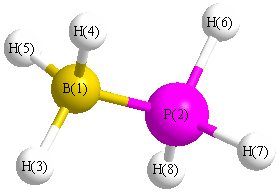 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| B1 |
P2 |
H6 |
117.675 |
|
B1 |
P2 |
H7 |
117.675 |
| B1 |
P2 |
H8 |
117.675 |
|
P2 |
B1 |
H3 |
103.603 |
| P2 |
B1 |
H4 |
103.603 |
|
P2 |
B1 |
H5 |
103.603 |
| H3 |
B1 |
H4 |
114.647 |
|
H3 |
B1 |
H5 |
114.647 |
| H4 |
B1 |
H5 |
114.647 |
|
H6 |
P2 |
H7 |
100.162 |
| H6 |
P2 |
H8 |
100.162 |
|
H7 |
P2 |
H8 |
100.162 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability