Jump to
S1C2
Energy calculated at B2PLYP=FULLultrafine/6-311G**
| | hartrees |
|---|
| Energy at 0K | -218.246736 |
| Energy at 298.15K | |
| HF Energy | -218.009717 |
| Nuclear repulsion energy | 127.953769 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at B2PLYP=FULLultrafine/6-311G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3134 |
3134 |
30.45 |
|
|
|
| 2 |
A' |
3068 |
3068 |
47.65 |
|
|
|
| 3 |
A' |
3057 |
3057 |
29.68 |
|
|
|
| 4 |
A' |
3052 |
3052 |
11.84 |
|
|
|
| 5 |
A' |
1538 |
1538 |
3.70 |
|
|
|
| 6 |
A' |
1524 |
1524 |
3.86 |
|
|
|
| 7 |
A' |
1510 |
1510 |
0.61 |
|
|
|
| 8 |
A' |
1454 |
1454 |
15.79 |
|
|
|
| 9 |
A' |
1432 |
1432 |
2.06 |
|
|
|
| 10 |
A' |
1351 |
1351 |
0.85 |
|
|
|
| 11 |
A' |
1153 |
1153 |
1.35 |
|
|
|
| 12 |
A' |
1075 |
1075 |
71.50 |
|
|
|
| 13 |
A' |
1046 |
1046 |
37.38 |
|
|
|
| 14 |
A' |
911 |
911 |
8.81 |
|
|
|
| 15 |
A' |
453 |
453 |
6.51 |
|
|
|
| 16 |
A' |
268 |
268 |
3.59 |
|
|
|
| 17 |
A" |
3130 |
3130 |
93.43 |
|
|
|
| 18 |
A" |
3111 |
3111 |
1.63 |
|
|
|
| 19 |
A" |
3093 |
3093 |
4.26 |
|
|
|
| 20 |
A" |
1517 |
1517 |
7.70 |
|
|
|
| 21 |
A" |
1333 |
1333 |
0.03 |
|
|
|
| 22 |
A" |
1286 |
1286 |
0.52 |
|
|
|
| 23 |
A" |
1209 |
1209 |
1.77 |
|
|
|
| 24 |
A" |
908 |
908 |
1.82 |
|
|
|
| 25 |
A" |
781 |
781 |
1.53 |
|
|
|
| 26 |
A" |
233 |
233 |
0.00 |
|
|
|
| 27 |
A" |
129 |
129 |
3.26 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21377.7 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 21377.7 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at B2PLYP=FULLultrafine/6-311G**
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.121 |
-0.784 |
0.000 |
| C2 |
0.000 |
0.725 |
0.000 |
| C3 |
-1.460 |
1.179 |
0.000 |
| F4 |
1.466 |
-1.156 |
0.000 |
| H5 |
-0.343 |
-1.218 |
0.890 |
| H6 |
-0.343 |
-1.218 |
-0.890 |
| H7 |
0.520 |
1.115 |
-0.879 |
| H8 |
0.520 |
1.115 |
0.879 |
| H9 |
-1.531 |
2.267 |
0.000 |
| H10 |
-1.990 |
0.811 |
-0.883 |
| H11 |
-1.990 |
0.811 |
0.883 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5143 | 2.5209 | 1.3947 | 1.0936 | 1.0936 | 2.1304 | 2.1304 | 3.4703 | 2.7896 | 2.7896 |
C2 | 1.5143 | | 1.5291 | 2.3843 | 2.1648 | 2.1648 | 1.0932 | 1.0932 | 2.1732 | 2.1785 | 2.1785 | C3 | 2.5209 | 1.5291 | | 3.7429 | 2.7905 | 2.7905 | 2.1675 | 2.1675 | 1.0910 | 1.0931 | 1.0931 | F4 | 1.3947 | 2.3843 | 3.7429 | | 2.0166 | 2.0166 | 2.6118 | 2.6118 | 4.5493 | 4.0728 | 4.0728 | H5 | 1.0936 | 2.1648 | 2.7905 | 2.0166 | | 1.7804 | 3.0525 | 2.4873 | 3.7887 | 3.1585 | 2.6137 | H6 | 1.0936 | 2.1648 | 2.7905 | 2.0166 | 1.7804 | | 2.4873 | 3.0525 | 3.7887 | 2.6137 | 3.1585 | H7 | 2.1304 | 1.0932 | 2.1675 | 2.6118 | 3.0525 | 2.4873 | | 1.7585 | 2.5115 | 2.5277 | 3.0814 | H8 | 2.1304 | 1.0932 | 2.1675 | 2.6118 | 2.4873 | 3.0525 | 1.7585 | | 2.5115 | 3.0814 | 2.5277 | H9 | 3.4703 | 2.1732 | 1.0910 | 4.5493 | 3.7887 | 3.7887 | 2.5115 | 2.5115 | | 1.7636 | 1.7636 | H10 | 2.7896 | 2.1785 | 1.0931 | 4.0728 | 3.1585 | 2.6137 | 2.5277 | 3.0814 | 1.7636 | | 1.7661 | H11 | 2.7896 | 2.1785 | 1.0931 | 4.0728 | 2.6137 | 3.1585 | 3.0814 | 2.5277 | 1.7636 | 1.7661 | |
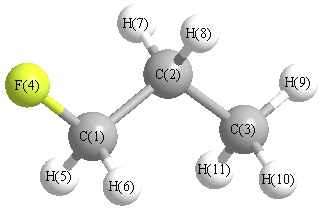 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
111.848 |
|
C1 |
C2 |
H7 |
108.494 |
| C1 |
C2 |
H8 |
108.494 |
|
C2 |
C1 |
F4 |
110.022 |
| C2 |
C1 |
H5 |
111.192 |
|
C2 |
C1 |
H6 |
111.192 |
| C2 |
C3 |
H9 |
110.973 |
|
C2 |
C3 |
H10 |
111.270 |
| C2 |
C3 |
H11 |
111.270 |
|
C3 |
C2 |
H7 |
110.386 |
| C3 |
C2 |
H8 |
110.386 |
|
F4 |
C1 |
H5 |
107.661 |
| F4 |
C1 |
H6 |
107.661 |
|
H5 |
C1 |
H6 |
108.982 |
| H7 |
C2 |
H8 |
107.085 |
|
H9 |
C3 |
H10 |
107.694 |
| H9 |
C3 |
H11 |
107.694 |
|
H10 |
C3 |
H11 |
107.770 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at B2PLYP=FULLultrafine/6-311G**
| | hartrees |
|---|
| Energy at 0K | -218.247446 |
| Energy at 298.15K | |
| HF Energy | -218.010274 |
| Nuclear repulsion energy | 130.416330 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at B2PLYP=FULLultrafine/6-311G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3145 |
3145 |
25.14 |
|
|
|
| 2 |
A |
3128 |
3128 |
58.25 |
|
|
|
| 3 |
A |
3117 |
3117 |
40.49 |
|
|
|
| 4 |
A |
3093 |
3093 |
18.07 |
|
|
|
| 5 |
A |
3062 |
3062 |
33.12 |
|
|
|
| 6 |
A |
3057 |
3057 |
27.77 |
|
|
|
| 7 |
A |
3052 |
3052 |
19.38 |
|
|
|
| 8 |
A |
1531 |
1531 |
1.01 |
|
|
|
| 9 |
A |
1526 |
1526 |
8.86 |
|
|
|
| 10 |
A |
1511 |
1511 |
7.19 |
|
|
|
| 11 |
A |
1490 |
1490 |
2.79 |
|
|
|
| 12 |
A |
1447 |
1447 |
11.16 |
|
|
|
| 13 |
A |
1428 |
1428 |
5.07 |
|
|
|
| 14 |
A |
1396 |
1396 |
0.41 |
|
|
|
| 15 |
A |
1317 |
1317 |
1.10 |
|
|
|
| 16 |
A |
1291 |
1291 |
1.29 |
|
|
|
| 17 |
A |
1192 |
1192 |
1.71 |
|
|
|
| 18 |
A |
1135 |
1135 |
8.53 |
|
|
|
| 19 |
A |
1096 |
1096 |
43.20 |
|
|
|
| 20 |
A |
988 |
988 |
49.66 |
|
|
|
| 21 |
A |
932 |
932 |
4.14 |
|
|
|
| 22 |
A |
886 |
886 |
4.33 |
|
|
|
| 23 |
A |
780 |
780 |
0.69 |
|
|
|
| 24 |
A |
484 |
484 |
4.33 |
|
|
|
| 25 |
A |
320 |
320 |
1.17 |
|
|
|
| 26 |
A |
225 |
225 |
1.90 |
|
|
|
| 27 |
A |
147 |
147 |
2.56 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21386.5 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 21386.5 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at B2PLYP=FULLultrafine/6-311G**
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.799 |
0.518 |
0.296 |
| C2 |
-0.594 |
0.661 |
-0.281 |
| C3 |
-1.521 |
-0.486 |
0.119 |
| F4 |
1.385 |
-0.663 |
-0.170 |
| H5 |
1.445 |
1.345 |
-0.005 |
| H6 |
0.772 |
0.459 |
1.387 |
| H7 |
-0.514 |
0.718 |
-1.371 |
| H8 |
-1.002 |
1.619 |
0.059 |
| H9 |
-2.507 |
-0.373 |
-0.336 |
| H10 |
-1.104 |
-1.441 |
-0.203 |
| H11 |
-1.653 |
-0.523 |
1.204 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5145 | 2.5341 | 1.3978 | 1.0923 | 1.0936 | 2.1306 | 2.1238 | 3.4817 | 2.7757 | 2.8137 |
C2 | 1.5145 | | 1.5281 | 2.3835 | 2.1687 | 2.1661 | 1.0938 | 1.0954 | 2.1754 | 2.1642 | 2.1743 | C3 | 2.5341 | 1.5281 | | 2.9260 | 3.4883 | 2.7860 | 2.1639 | 2.1692 | 1.0915 | 1.0906 | 1.0934 | F4 | 1.3978 | 2.3835 | 2.9260 | | 2.0156 | 2.0148 | 2.6366 | 3.3098 | 3.9064 | 2.6080 | 3.3367 | H5 | 1.0923 | 2.1687 | 3.4883 | 2.0156 | | 1.7826 | 2.4690 | 2.4628 | 4.3225 | 3.7815 | 3.8142 | H6 | 1.0936 | 2.1661 | 2.7860 | 2.0148 | 1.7826 | | 3.0541 | 2.5016 | 3.7968 | 3.1074 | 2.6225 | H7 | 2.1306 | 1.0938 | 2.1639 | 2.6366 | 2.4690 | 3.0541 | | 1.7589 | 2.4970 | 2.5245 | 3.0764 | H8 | 2.1238 | 1.0954 | 2.1692 | 3.3098 | 2.4628 | 2.5016 | 1.7589 | | 2.5284 | 3.0730 | 2.5150 | H9 | 3.4817 | 2.1754 | 1.0915 | 3.9064 | 4.3225 | 3.7968 | 2.4970 | 2.5284 | | 1.7682 | 1.7672 | H10 | 2.7757 | 2.1642 | 1.0906 | 2.6080 | 3.7815 | 3.1074 | 2.5245 | 3.0730 | 1.7682 | | 1.7663 | H11 | 2.8137 | 2.1743 | 1.0934 | 3.3367 | 3.8142 | 2.6225 | 3.0764 | 2.5150 | 1.7672 | 1.7663 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
112.791 |
|
C1 |
C2 |
H7 |
108.466 |
| C1 |
C2 |
H8 |
107.849 |
|
C2 |
C1 |
F4 |
109.792 |
| C2 |
C1 |
H5 |
111.576 |
|
C2 |
C1 |
H6 |
111.284 |
| C2 |
C3 |
H9 |
111.196 |
|
C2 |
C3 |
H10 |
110.358 |
| C2 |
C3 |
H11 |
110.996 |
|
C3 |
C2 |
H7 |
110.140 |
| C3 |
C2 |
H8 |
110.467 |
|
F4 |
C1 |
H5 |
107.447 |
| F4 |
C1 |
H6 |
107.302 |
|
H5 |
C1 |
H6 |
109.274 |
| H7 |
C2 |
H8 |
106.920 |
|
H9 |
C3 |
H10 |
108.259 |
| H9 |
C3 |
H11 |
107.967 |
|
H10 |
C3 |
H11 |
107.946 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
