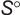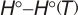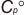Geometric Data
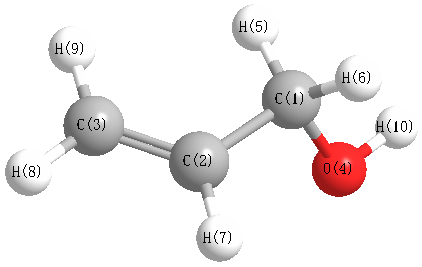
Point Group C1
Internal coordinates
distances (r) in Å, angles (a) in degrees, dihedrals (d) in degrees
Cartesians
| Atom |
x (Å) |
y (Å) |
z (Å) |
| C1 |
-0.5612 |
-0.5724 |
0.1206 |
| C2 |
0.7021 |
0.1891 |
0.4094 |
| C3 |
1.8384 |
0.0528 |
-0.2727 |
| O4 |
-1.6567 |
0.2923 |
-0.1596 |
| H5 |
-0.2958 |
-1.2779 |
-0.7135 |
| H6 |
-0.8201 |
-1.1509 |
1.0140 |
| H7 |
0.6253 |
0.9264 |
1.2199 |
| H8 |
1.8874 |
-0.6738 |
-1.0965 |
| H9 |
2.7226 |
0.6206 |
-0.0601 |
| H10 |
-1.3939 |
0.8550 |
-0.8945 |
Atom - Atom Distances 
Distances in Å
| |
C1 |
C2 |
C3 |
O4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
| C1 |
|
1.5031 | 2.5107 | 1.4235 | 1.1242 | 1.0954 | 2.2051 | 2.7363 | 3.4985 | 1.9394 |
| C2 |
1.5031 |
|
1.3323 | 2.4287 | 2.0997 | 2.1162 | 1.0984 | 2.1017 | 2.1187 | 2.5567 |
| C3 |
2.5107 | 1.3323 |
|
3.5051 | 2.5534 | 3.1894 | 2.1125 | 1.0995 | 1.0721 | 3.3879 |
| O4 |
1.4235 | 2.4287 | 3.5051 |
|
2.1504 | 2.0396 | 2.7409 | 3.7910 | 4.3927 | 0.9622 |
| H5 |
1.1242 | 2.0997 | 2.5534 | 2.1504 |
|
1.8098 | 3.0733 | 2.2974 | 3.6252 | 2.4058 |
| H6 |
1.0954 | 2.1162 | 3.1894 | 2.0396 | 1.8098 |
|
2.5390 | 3.4659 | 4.1040 | 2.8276 |
| H7 |
2.2051 | 1.0984 | 2.1125 | 2.7409 | 3.0733 | 2.5390 |
|
3.0853 | 2.4760 | 2.9245 |
| H8 |
2.7363 | 2.1017 | 1.0995 | 3.7910 | 2.2974 | 3.4659 | 3.0853 |
|
1.8567 | 3.6256 |
| H9 |
3.4985 | 2.1187 | 1.0721 | 4.3927 | 3.6252 | 4.1040 | 2.4760 | 1.8567 |
|
4.2067 |
| H10 |
1.9394 | 2.5567 | 3.3879 | 0.9622 | 2.4058 | 2.8276 | 2.9245 | 3.6256 | 4.2067 |
|
Calculated geometries
for C
3H
6O (2-Propen-1-ol).
Experimental Bond Angles (degrees) from cartesians 
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
124.516 |
|
C1 |
C2 |
H7 |
115.030 |
| C1 |
O4 |
H10 |
107.181 |
|
C2 |
C1 |
O4 |
112.141 |
| C2 |
C1 |
H5 |
105.193 |
|
C2 |
C1 |
H6 |
108.030 |
| C2 |
C3 |
H8 |
119.288 |
|
C2 |
C3 |
H9 |
123.209 |
| C3 |
C2 |
H7 |
120.404 |
|
O4 |
C1 |
H5 |
114.635 |
| O4 |
C1 |
H6 |
107.425 |
|
H5 |
C1 |
H6 |
109.238 |
| H8 |
C3 |
H9 |
117.503 |
Bond descriptions
Examples: C-C single bond, C=C, double bond, C#C triple bond, C:C aromatic bond
| Bond Type |
Count |
| H-C |
5 |
| H-O |
1 |
| C-C |
1 |
| C=C |
1 |
| C-O |
1 |
Connectivity
| Atom 1 |
Atom 2 |
| C1 |
C2 |
| C1 |
O4 |
| C1 |
H5 |
| C1 |
H6 |
| C2 |
C3 |
| C2 |
H7 |
| C3 |
H8 |
| C3 |
H9 |
| O4 |
H10 |