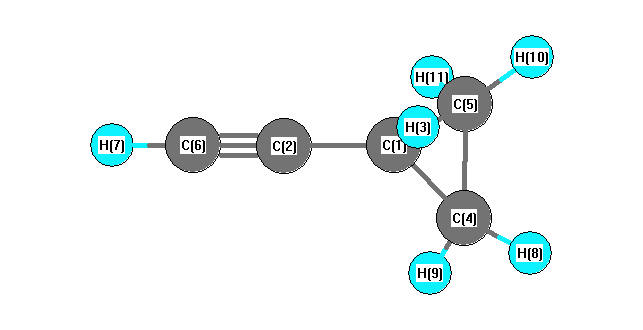
Geometric Data
Point Group Cs
Internal coordinates
distances (r) in Å, angles (a) in degrees, dihedrals (d) in degrees
Cartesians
| Atom |
x (Å) |
y (Å) |
z (Å) |
| C1 |
-0.2523 |
0.0000 |
0.5778 |
| C2 |
1.1078 |
0.0000 |
0.1626 |
| H3 |
-0.3254 |
0.0000 |
1.6553 |
| C4 |
-1.3015 |
0.7519 |
-0.2372 |
| C5 |
-1.3015 |
-0.7519 |
-0.2372 |
| C6 |
2.2669 |
0.0000 |
-0.1881 |
| H7 |
3.2764 |
0.0000 |
-0.4944 |
| H8 |
-2.0662 |
1.2729 |
0.3108 |
| H9 |
-0.9672 |
1.2571 |
-1.1247 |
| H10 |
-2.0662 |
-1.2729 |
0.3108 |
| H11 |
-0.9672 |
-1.2571 |
-1.1247 |
Atom - Atom Distances 
Distances in Å
| |
C1 |
C2 |
H3 |
C4 |
C5 |
C6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 |
|
1.4220 | 1.0800 | 1.5266 | 1.5266 | 2.6330 | 3.6880 | 2.2320 | 2.2339 | 2.2320 | 2.2339 |
| C2 |
1.4220 |
|
2.0694 | 2.5554 | 2.5554 | 1.2110 | 2.2660 | 3.4229 | 2.7465 | 3.4229 | 2.7465 |
| H3 |
1.0800 | 2.0694 |
|
2.2583 | 2.2583 | 3.1809 | 4.1946 | 2.5413 | 3.1179 | 2.5413 | 3.1179 |
| C4 |
1.5266 | 2.5554 | 2.2583 |
|
1.5038 | 3.6471 | 4.6464 | 1.0754 | 1.0746 | 2.2326 | 2.2216 |
| C5 |
1.5266 | 2.5554 | 2.2583 | 1.5038 |
|
3.6471 | 4.6464 | 2.2326 | 2.2216 | 1.0754 | 1.0746 |
| C6 |
2.6330 | 1.2110 | 3.1809 | 3.6471 | 3.6471 |
|
1.0550 | 4.5436 | 3.5941 | 4.5436 | 3.5941 |
| H7 |
3.6880 | 2.2660 | 4.1946 | 4.6464 | 4.6464 | 1.0550 |
|
5.5509 | 4.4706 | 5.5509 | 4.4706 |
| H8 |
2.2320 | 3.4229 | 2.5413 | 1.0754 | 2.2326 | 4.5436 | 5.5509 |
|
1.8080 | 2.5457 | 3.1096 |
| H9 |
2.2339 | 2.7465 | 3.1179 | 1.0746 | 2.2216 | 3.5941 | 4.4706 | 1.8080 |
|
3.1096 | 2.5143 |
| H10 |
2.2320 | 3.4229 | 2.5413 | 2.2326 | 1.0754 | 4.5436 | 5.5509 | 2.5457 | 3.1096 |
|
1.8080 |
| H11 |
2.2339 | 2.7465 | 3.1179 | 2.2216 | 1.0746 | 3.5941 | 4.4706 | 3.1096 | 2.5143 | 1.8080 |
|
Calculated geometries
for C
5H
6 (Cyclopropylacetylene).
Experimental Bond Angles (degrees) from cartesians 
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C6 |
179.852 |
|
C1 |
C4 |
C5 |
60.494 |
| C1 |
C4 |
H8 |
117.078 |
|
C1 |
C4 |
H9 |
117.303 |
| C1 |
C5 |
C4 |
60.494 |
|
C1 |
C5 |
H10 |
117.078 |
| C1 |
C5 |
H11 |
117.303 |
|
C2 |
C1 |
H3 |
110.863 |
| C2 |
C1 |
C4 |
120.096 |
|
C2 |
C1 |
C5 |
120.096 |
| C2 |
C6 |
H7 |
179.953 |
|
H3 |
C1 |
C4 |
119.085 |
| H3 |
C1 |
C5 |
119.085 |
|
C4 |
C1 |
C5 |
59.011 |
| C4 |
C5 |
H10 |
118.976 |
|
C4 |
C5 |
H11 |
118.047 |
| C5 |
C4 |
H8 |
118.976 |
|
C5 |
C4 |
H9 |
118.047 |
| H8 |
C4 |
H9 |
114.478 |
|
H10 |
C5 |
H11 |
114.478 |
Bond descriptions
Examples: C-C single bond, C=C, double bond, C#C triple bond, C:C aromatic bond
| Bond Type |
Count |
| H-C |
6 |
| C-C |
4 |
| C#C |
1 |
Connectivity
| Atom 1 |
Atom 2 |
| C1 |
C2 |
| C1 |
H3 |
| C1 |
C4 |
| C1 |
C5 |
| C2 |
C6 |
| C4 |
C5 |
| C4 |
H8 |
| C4 |
H9 |
| C5 |
H10 |
| C5 |
H11 |
| C6 |
H7 |