|
|
II.A.3. (XII.A.1.) |
Listing of experimental geometry data for NH2OH (hydroxylamine)
Rotational Constants (cm-1)
See section I.F.4 to change rotational constant units
| A | B | C |
|---|---|---|
| 6.37031 | 0.84124 | 0.83911 |
Calculated rotational constants for NH2OH (hydroxylamine).
Point Group Cs
Internal coordinates
(distances (r) in Å) (angles (a) in degrees) (dihedrals (d) in degrees)| Description | Value | Connectivity | Reference | Comment | |||
|---|---|---|---|---|---|---|---|
| Atom 1 | Atom 2 | Atom 3 | Atom 4 | ||||
| rOH | 0.962 | 2 | 3 | 1972Tsu:167-174 | |||
| rNH | 1.016 | 1 | 4 | 1972Tsu:167-174 | |||
| rNO | 1.453 | 1 | 2 | 1972Tsu:167-174 | |||
| aHON | 101.37 | 1 | 2 | 3 | 1972Tsu:167-174 | ||
| aHNO | 107.01 | 2 | 1 | 4 | 1972Tsu:167-174 | ||
| aHNH | 103.25 | 4 | 1 | 5 | 1972Tsu:167-174 | ||
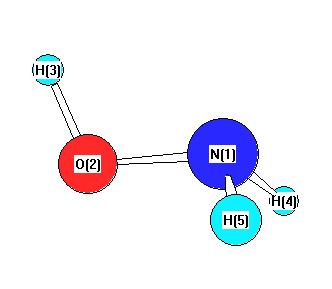
Bond descriptions
Examples: C-C single bond, C=C, double bond, C#C triple bond, C:C aromatic bond
| Bond Type | Count |
|---|---|
| N-O | 1 |
| H-N | 2 |
| H-O | 1 |
| Atom | x (Å) | y (Å) | z (Å) |
|---|---|---|---|
| N1 | -0.0094 | 0.7040 | 0.0000 |
| O2 | -0.0094 | -0.7490 | 0.0000 |
| H3 | -0.9525 | -0.9386 | 0.0000 |
| H4 | 0.5469 | 1.0012 | 0.7965 |
| H5 | 0.5469 | 1.0012 | -0.7965 |
Atom - Atom Distances (Å)
| N1 | O2 | H3 | H4 | H5 | |
|---|---|---|---|---|---|
| N1 | 1.4530 | 1.8941 | 1.0160 | 1.0160 | |
| O2 | 1.4530 | 0.9620 | 2.0018 | 2.0018 | |
| H3 | 1.8941 | 0.9620 | 2.5780 | 2.5780 | |
| H4 | 1.0160 | 2.0018 | 2.5780 | 1.5930 | |
| H5 | 1.0160 | 2.0018 | 2.5780 | 1.5930 |
Calculated geometries for NH2OH (hydroxylamine).
References
By selecting the following links, you may be leaving NIST webspace. We have provided these links to other web sites because they may have information that would be of interest to you. No inferences should be drawn on account of other sites being referenced, or not, from this page. There may be other web sites that are more appropriate for your purpose. NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites. Further, NIST does not endorse any commercial products that may be mentioned on these sites. Please address comments about this page to cccbdb@nist.gov.
By selecting the following links, you may be leaving NIST webspace. We have provided these links to other web sites because they may have information that would be of interest to you. No inferences should be drawn on account of other sites being referenced, or not, from this page. There may be other web sites that are more appropriate for your purpose. NIST does not necessarily endorse the views expressed, or concur with the facts presented on these sites. Further, NIST does not endorse any commercial products that may be mentioned on these sites. Please address comments about this page to cccbdb@nist.gov.
| squib | reference | DOI |
|---|---|---|
| 1972Tsu:167-174 | S Tsunekawa "Microwave Spectrum of Hydroxylamine" J. Phys. Soc. Japan 33(1), 167-174, 1972 | 10.1143/JPSJ.33.167 |
Got a better number? Please email us at
cccbdb@nist.gov
| Browse | |
|---|---|
| Previous | Next |