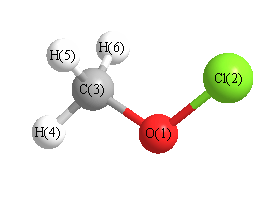
Vibrational Frequencies calculated at LSDA/6-311G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3040 |
3003 |
5.95 |
|
|
|
| 2 |
A' |
2925 |
2889 |
28.96 |
|
|
|
| 3 |
A' |
1441 |
1423 |
13.54 |
|
|
|
| 4 |
A' |
1380 |
1363 |
0.36 |
|
|
|
| 5 |
A' |
1162 |
1147 |
24.62 |
|
|
|
| 6 |
A' |
1026 |
1013 |
20.03 |
|
|
|
| 7 |
A' |
649 |
641 |
4.35 |
|
|
|
| 8 |
A' |
358 |
354 |
5.67 |
|
|
|
| 9 |
A" |
2996 |
2959 |
30.91 |
|
|
|
| 10 |
A" |
1368 |
1351 |
13.87 |
|
|
|
| 11 |
A" |
1134 |
1120 |
0.02 |
|
|
|
| 12 |
A" |
280 |
276 |
5.10 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 8878.6 cm
-1
Scaled (by 0.9877) Zero Point Vibrational Energy (zpe) 8769.4 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at LSDA/6-311G**
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
O |
-0.360 |
|
|
|
| 2 |
Cl |
0.115 |
|
|
|
| 3 |
C |
-0.257 |
|
|
|
| 4 |
H |
0.176 |
|
|
|
| 5 |
H |
0.163 |
|
|
|
| 6 |
H |
0.163 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
1.919 |
-0.149 |
0.000 |
1.925 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-22.899 |
2.286 |
0.000 |
| y |
2.286 |
-24.508 |
0.000 |
| z |
0.000 |
0.000 |
-25.843 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
2.276 |
2.286 |
0.000 |
| y |
2.286 |
-0.137 |
0.000 |
| z |
0.000 |
0.000 |
-2.139 |
|
| Polar |
|---|
| 3z2-r2 | -4.277 |
|---|
| x2-y2 | 1.609 |
|---|
| xy | 2.286 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
4.884 |
1.453 |
0.000 |
| y |
1.453 |
4.436 |
0.000 |
| z |
0.000 |
0.000 |
2.916 |
<r2> (average value of r
2) Å
2
| <r2> |
67.549 |
| (<r2>)1/2 |
8.219 |