Jump to
S1C2
Energy calculated at MP2=FULL/6-311+G(3df,2p)
| | hartrees |
|---|
| Energy at 0K | -218.057536 |
| Energy at 298.15K | -218.065424 |
| HF Energy | -217.193353 |
| Nuclear repulsion energy | 128.713178 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2=FULL/6-311+G(3df,2p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3181 |
2998 |
18.59 |
|
|
|
| 2 |
A' |
3100 |
2922 |
38.74 |
|
|
|
| 3 |
A' |
3088 |
2911 |
18.87 |
|
|
|
| 4 |
A' |
3083 |
2906 |
10.98 |
|
|
|
| 5 |
A' |
1544 |
1456 |
5.05 |
|
|
|
| 6 |
A' |
1530 |
1443 |
4.93 |
|
|
|
| 7 |
A' |
1515 |
1428 |
0.34 |
|
|
|
| 8 |
A' |
1444 |
1361 |
14.46 |
|
|
|
| 9 |
A' |
1426 |
1345 |
1.32 |
|
|
|
| 10 |
A' |
1346 |
1268 |
0.55 |
|
|
|
| 11 |
A' |
1160 |
1093 |
1.39 |
|
|
|
| 12 |
A' |
1092 |
1029 |
78.49 |
|
|
|
| 13 |
A' |
1064 |
1003 |
40.43 |
|
|
|
| 14 |
A' |
918 |
866 |
7.68 |
|
|
|
| 15 |
A' |
459 |
433 |
6.55 |
|
|
|
| 16 |
A' |
269 |
254 |
3.46 |
|
|
|
| 17 |
A" |
3174 |
2992 |
52.43 |
|
|
|
| 18 |
A" |
3157 |
2976 |
3.02 |
|
|
|
| 19 |
A" |
3138 |
2958 |
2.87 |
|
|
|
| 20 |
A" |
1526 |
1438 |
7.55 |
|
|
|
| 21 |
A" |
1336 |
1259 |
0.01 |
|
|
|
| 22 |
A" |
1290 |
1216 |
0.62 |
|
|
|
| 23 |
A" |
1209 |
1140 |
0.99 |
|
|
|
| 24 |
A" |
907 |
855 |
1.09 |
|
|
|
| 25 |
A" |
774 |
730 |
1.78 |
|
|
|
| 26 |
A" |
238 |
224 |
0.01 |
|
|
|
| 27 |
A" |
136 |
128 |
3.41 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21551.0 cm
-1
Scaled (by 0.9427) Zero Point Vibrational Energy (zpe) 20316.1 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2=FULL/6-311+G(3df,2p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.128 |
-0.768 |
0.000 |
| C2 |
0.000 |
0.732 |
0.000 |
| C3 |
-1.462 |
1.145 |
0.000 |
| F4 |
1.464 |
-1.140 |
0.000 |
| H5 |
-0.333 |
-1.202 |
0.887 |
| H6 |
-0.333 |
-1.202 |
-0.887 |
| H7 |
0.510 |
1.128 |
-0.877 |
| H8 |
0.510 |
1.128 |
0.877 |
| H9 |
-1.563 |
2.227 |
0.000 |
| H10 |
-1.979 |
0.765 |
-0.880 |
| H11 |
-1.979 |
0.765 |
0.880 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5050 | 2.4877 | 1.3875 | 1.0897 | 1.0897 | 2.1235 | 2.1235 | 3.4391 | 2.7500 | 2.7500 |
C2 | 1.5050 | | 1.5198 | 2.3765 | 2.1535 | 2.1535 | 1.0890 | 1.0890 | 2.1630 | 2.1659 | 2.1659 | C3 | 2.4877 | 1.5198 | | 3.7134 | 2.7519 | 2.7519 | 2.1583 | 2.1583 | 1.0864 | 1.0885 | 1.0885 | F4 | 1.3875 | 2.3765 | 3.7134 | | 2.0053 | 2.0053 | 2.6125 | 2.6125 | 4.5279 | 4.0325 | 4.0325 | H5 | 1.0897 | 2.1535 | 2.7519 | 2.0053 | | 1.7732 | 3.0416 | 2.4781 | 3.7495 | 3.1145 | 2.5653 | H6 | 1.0897 | 2.1535 | 2.7519 | 2.0053 | 1.7732 | | 2.4781 | 3.0416 | 3.7495 | 2.5653 | 3.1145 | H7 | 2.1235 | 1.0890 | 2.1583 | 2.6125 | 3.0416 | 2.4781 | | 1.7540 | 2.5044 | 2.5148 | 3.0676 | H8 | 2.1235 | 1.0890 | 2.1583 | 2.6125 | 2.4781 | 3.0416 | 1.7540 | | 2.5044 | 3.0676 | 2.5148 | H9 | 3.4391 | 2.1630 | 1.0864 | 4.5279 | 3.7495 | 3.7495 | 2.5044 | 2.5044 | | 1.7559 | 1.7559 | H10 | 2.7500 | 2.1659 | 1.0885 | 4.0325 | 3.1145 | 2.5653 | 2.5148 | 3.0676 | 1.7559 | | 1.7592 | H11 | 2.7500 | 2.1659 | 1.0885 | 4.0325 | 2.5653 | 3.1145 | 3.0676 | 2.5148 | 1.7559 | 1.7592 | |
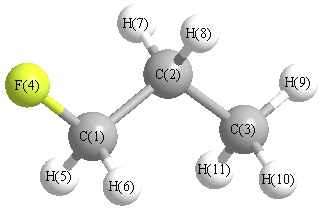 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
110.660 |
|
C1 |
C2 |
H7 |
108.839 |
| C1 |
C2 |
H8 |
108.839 |
|
C2 |
C1 |
F4 |
110.431 |
| C2 |
C1 |
H5 |
111.188 |
|
C2 |
C1 |
H6 |
111.188 |
| C2 |
C3 |
H9 |
111.094 |
|
C2 |
C3 |
H10 |
111.200 |
| C2 |
C3 |
H11 |
111.200 |
|
C3 |
C2 |
H7 |
110.562 |
| C3 |
C2 |
H8 |
110.562 |
|
F4 |
C1 |
H5 |
107.488 |
| F4 |
C1 |
H6 |
107.488 |
|
H5 |
C1 |
H6 |
108.910 |
| H7 |
C2 |
H8 |
107.290 |
|
H9 |
C3 |
H10 |
107.679 |
| H9 |
C3 |
H11 |
107.679 |
|
H10 |
C3 |
H11 |
107.820 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at MP2=FULL/6-311+G(3df,2p)
| | hartrees |
|---|
| Energy at 0K | -218.057894 |
| Energy at 298.15K | |
| HF Energy | -217.193373 |
| Nuclear repulsion energy | 131.122321 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2=FULL/6-311+G(3df,2p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3188 |
3005 |
14.63 |
|
|
|
| 2 |
A |
3173 |
2991 |
32.08 |
|
|
|
| 3 |
A |
3160 |
2979 |
28.52 |
|
|
|
| 4 |
A |
3137 |
2957 |
10.95 |
|
|
|
| 5 |
A |
3094 |
2917 |
32.98 |
|
|
|
| 6 |
A |
3087 |
2910 |
22.54 |
|
|
|
| 7 |
A |
3084 |
2907 |
9.95 |
|
|
|
| 8 |
A |
1539 |
1451 |
2.65 |
|
|
|
| 9 |
A |
1532 |
1444 |
8.42 |
|
|
|
| 10 |
A |
1520 |
1433 |
6.68 |
|
|
|
| 11 |
A |
1493 |
1408 |
3.63 |
|
|
|
| 12 |
A |
1442 |
1359 |
9.66 |
|
|
|
| 13 |
A |
1425 |
1344 |
4.33 |
|
|
|
| 14 |
A |
1392 |
1313 |
0.04 |
|
|
|
| 15 |
A |
1318 |
1243 |
1.68 |
|
|
|
| 16 |
A |
1295 |
1221 |
1.42 |
|
|
|
| 17 |
A |
1193 |
1125 |
1.27 |
|
|
|
| 18 |
A |
1140 |
1075 |
8.60 |
|
|
|
| 19 |
A |
1115 |
1051 |
51.38 |
|
|
|
| 20 |
A |
996 |
939 |
47.72 |
|
|
|
| 21 |
A |
934 |
880 |
2.51 |
|
|
|
| 22 |
A |
902 |
850 |
4.50 |
|
|
|
| 23 |
A |
778 |
733 |
0.64 |
|
|
|
| 24 |
A |
486 |
458 |
4.15 |
|
|
|
| 25 |
A |
324 |
305 |
1.15 |
|
|
|
| 26 |
A |
227 |
214 |
1.77 |
|
|
|
| 27 |
A |
147 |
138 |
2.38 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21558.5 cm
-1
Scaled (by 0.9427) Zero Point Vibrational Energy (zpe) 20323.2 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2=FULL/6-311+G(3df,2p)
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.787 |
0.511 |
0.301 |
| C2 |
-0.591 |
0.654 |
-0.287 |
| C3 |
-1.513 |
-0.483 |
0.118 |
| F4 |
1.385 |
-0.651 |
-0.173 |
| H5 |
1.433 |
1.342 |
0.024 |
| H6 |
0.748 |
0.432 |
1.387 |
| H7 |
-0.504 |
0.709 |
-1.371 |
| H8 |
-0.994 |
1.611 |
0.049 |
| H9 |
-2.503 |
-0.358 |
-0.311 |
| H10 |
-1.117 |
-1.437 |
-0.218 |
| H11 |
-1.621 |
-0.527 |
1.201 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5048 | 2.5117 | 1.3899 | 1.0883 | 1.0898 | 2.1219 | 2.1087 | 3.4577 | 2.7728 | 2.7717 |
C2 | 1.5048 | | 1.5184 | 2.3706 | 2.1600 | 2.1549 | 1.0896 | 1.0912 | 2.1638 | 2.1575 | 2.1600 | C3 | 2.5117 | 1.5184 | | 2.9167 | 3.4661 | 2.7493 | 2.1579 | 2.1579 | 1.0869 | 1.0865 | 1.0888 | F4 | 1.3899 | 2.3706 | 2.9167 | | 2.0035 | 2.0033 | 2.6181 | 3.2902 | 3.9014 | 2.6225 | 3.3067 | H5 | 1.0883 | 2.1600 | 3.4661 | 2.0035 | | 1.7758 | 2.4698 | 2.4421 | 4.3007 | 3.7793 | 3.7680 | H6 | 1.0898 | 2.1549 | 2.7493 | 2.0033 | 1.7758 | | 3.0420 | 2.4931 | 3.7525 | 3.0901 | 2.5622 | H7 | 2.1219 | 1.0896 | 2.1579 | 2.6181 | 2.4698 | 3.0420 | | 1.7522 | 2.5019 | 2.5127 | 3.0641 | H8 | 2.1087 | 1.0912 | 2.1579 | 3.2902 | 2.4421 | 2.4931 | 1.7522 | | 2.5068 | 3.0621 | 2.5073 | H9 | 3.4577 | 2.1638 | 1.0869 | 3.9014 | 4.3007 | 3.7525 | 2.5019 | 2.5068 | | 1.7593 | 1.7589 | H10 | 2.7728 | 2.1575 | 1.0865 | 2.6225 | 3.7793 | 3.0901 | 2.5127 | 3.0621 | 1.7593 | | 1.7592 | H11 | 2.7717 | 2.1600 | 1.0888 | 3.3067 | 3.7680 | 2.5622 | 3.0641 | 2.5073 | 1.7589 | 1.7592 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
112.366 |
|
C1 |
C2 |
H7 |
108.689 |
| C1 |
C2 |
H8 |
107.572 |
|
C2 |
C1 |
F4 |
109.893 |
| C2 |
C1 |
H5 |
111.810 |
|
C2 |
C1 |
H6 |
111.301 |
| C2 |
C3 |
H9 |
111.230 |
|
C2 |
C3 |
H10 |
110.748 |
| C2 |
C3 |
H11 |
110.810 |
|
C3 |
C2 |
H7 |
110.592 |
| C3 |
C2 |
H8 |
110.498 |
|
F4 |
C1 |
H5 |
107.262 |
| F4 |
C1 |
H6 |
107.154 |
|
H5 |
C1 |
H6 |
109.231 |
| H7 |
C2 |
H8 |
106.923 |
|
H9 |
C3 |
H10 |
108.089 |
| H9 |
C3 |
H11 |
107.885 |
|
H10 |
C3 |
H11 |
107.950 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
