Jump to
S1C2
Energy calculated at MP2=FULL/6-31G**
| | hartrees |
|---|
| Energy at 0K | -193.768975 |
| Energy at 298.15K | -193.777992 |
| Nuclear repulsion energy | 130.825034 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2=FULL/6-31G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3900 |
3644 |
20.81 |
|
|
|
| 2 |
A' |
3231 |
3019 |
26.21 |
|
|
|
| 3 |
A' |
3147 |
2940 |
22.94 |
|
|
|
| 4 |
A' |
3129 |
2924 |
21.30 |
|
|
|
| 5 |
A' |
3069 |
2867 |
54.61 |
|
|
|
| 6 |
A' |
1592 |
1488 |
3.85 |
|
|
|
| 7 |
A' |
1571 |
1468 |
4.35 |
|
|
|
| 8 |
A' |
1555 |
1453 |
0.60 |
|
|
|
| 9 |
A' |
1508 |
1409 |
7.00 |
|
|
|
| 10 |
A' |
1470 |
1373 |
0.85 |
|
|
|
| 11 |
A' |
1387 |
1296 |
4.89 |
|
|
|
| 12 |
A' |
1289 |
1205 |
56.38 |
|
|
|
| 13 |
A' |
1135 |
1061 |
6.58 |
|
|
|
| 14 |
A' |
1106 |
1034 |
64.73 |
|
|
|
| 15 |
A' |
1084 |
1013 |
27.14 |
|
|
|
| 16 |
A' |
922 |
861 |
5.22 |
|
|
|
| 17 |
A' |
468 |
437 |
10.77 |
|
|
|
| 18 |
A' |
280 |
261 |
4.53 |
|
|
|
| 19 |
A" |
3224 |
3013 |
52.51 |
|
|
|
| 20 |
A" |
3201 |
2991 |
4.87 |
|
|
|
| 21 |
A" |
3116 |
2911 |
56.43 |
|
|
|
| 22 |
A" |
1564 |
1462 |
5.99 |
|
|
|
| 23 |
A" |
1361 |
1272 |
0.31 |
|
|
|
| 24 |
A" |
1309 |
1223 |
0.01 |
|
|
|
| 25 |
A" |
1226 |
1146 |
1.79 |
|
|
|
| 26 |
A" |
930 |
869 |
2.84 |
|
|
|
| 27 |
A" |
787 |
736 |
0.31 |
|
|
|
| 28 |
A" |
289 |
270 |
122.68 |
|
|
|
| 29 |
A" |
246 |
230 |
5.64 |
|
|
|
| 30 |
A" |
133 |
124 |
5.66 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 24614.2 cm
-1
Scaled (by 0.9344) Zero Point Vibrational Energy (zpe) 22999.5 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2=FULL/6-31G**
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-1.441 |
1.227 |
0.000 |
| C2 |
0.000 |
0.736 |
0.000 |
| C3 |
0.092 |
-0.774 |
0.000 |
| O4 |
1.472 |
-1.123 |
0.000 |
| H5 |
-1.485 |
2.314 |
0.000 |
| H6 |
-1.976 |
0.873 |
0.880 |
| H7 |
-1.976 |
0.873 |
-0.880 |
| H8 |
0.530 |
1.110 |
0.876 |
| H9 |
0.530 |
1.110 |
-0.876 |
| H10 |
-0.419 |
-1.175 |
0.883 |
| H11 |
-0.419 |
-1.175 |
-0.883 |
| H12 |
1.531 |
-2.084 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
O4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
H12 |
| C1 | | 1.5224 | 2.5206 | 3.7429 | 1.0874 | 1.0890 | 1.0890 | 2.1603 | 2.1603 | 2.7556 | 2.7556 | 4.4494 |
C2 | 1.5224 | | 1.5130 | 2.3715 | 2.1662 | 2.1673 | 2.1673 | 1.0897 | 1.0897 | 2.1464 | 2.1464 | 3.2093 | C3 | 2.5206 | 1.5130 | | 1.4238 | 3.4667 | 2.7859 | 2.7859 | 2.1232 | 2.1232 | 1.0959 | 1.0959 | 1.9463 | O4 | 3.7429 | 2.3715 | 1.4238 | | 4.5336 | 4.0800 | 4.0800 | 2.5768 | 2.5768 | 2.0875 | 2.0875 | 0.9632 | H5 | 1.0874 | 2.1662 | 3.4667 | 4.5336 | | 1.7583 | 1.7583 | 2.5051 | 2.5051 | 3.7529 | 3.7529 | 5.3325 | H6 | 1.0890 | 2.1673 | 2.7859 | 4.0800 | 1.7583 | | 1.7601 | 2.5174 | 3.0691 | 2.5723 | 3.1185 | 4.6707 | H7 | 1.0890 | 2.1673 | 2.7859 | 4.0800 | 1.7583 | 1.7601 | | 3.0691 | 2.5174 | 3.1185 | 2.5723 | 4.6707 | H8 | 2.1603 | 1.0897 | 2.1232 | 2.5768 | 2.5051 | 2.5174 | 3.0691 | | 1.7510 | 2.4740 | 3.0353 | 3.4600 | H9 | 2.1603 | 1.0897 | 2.1232 | 2.5768 | 2.5051 | 3.0691 | 2.5174 | 1.7510 | | 3.0353 | 2.4740 | 3.4600 | H10 | 2.7556 | 2.1464 | 1.0959 | 2.0875 | 3.7529 | 2.5723 | 3.1185 | 2.4740 | 3.0353 | | 1.7659 | 2.3254 | H11 | 2.7556 | 2.1464 | 1.0959 | 2.0875 | 3.7529 | 3.1185 | 2.5723 | 3.0353 | 2.4740 | 1.7659 | | 2.3254 | H12 | 4.4494 | 3.2093 | 1.9463 | 0.9632 | 5.3325 | 4.6707 | 4.6707 | 3.4600 | 3.4600 | 2.3254 | 2.3254 | |
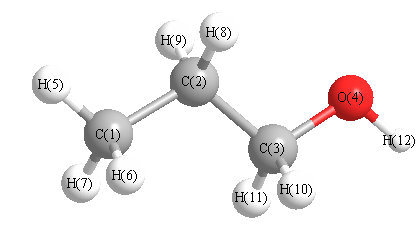 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
112.284 |
|
C1 |
C2 |
H8 |
110.496 |
| C1 |
C2 |
H9 |
110.496 |
|
C2 |
C1 |
H5 |
111.105 |
| C2 |
C1 |
H6 |
111.099 |
|
C2 |
C1 |
H7 |
111.099 |
| C2 |
C3 |
O4 |
107.671 |
|
C2 |
C3 |
H10 |
109.683 |
| C2 |
C3 |
H11 |
109.683 |
|
C3 |
C2 |
H8 |
108.228 |
| C3 |
C2 |
H9 |
108.228 |
|
C3 |
O4 |
H12 |
107.680 |
| O4 |
C3 |
H10 |
111.222 |
|
O4 |
C3 |
H11 |
111.222 |
| H5 |
C1 |
H6 |
107.777 |
|
H5 |
C1 |
H7 |
107.777 |
| H6 |
C1 |
H7 |
107.825 |
|
H8 |
C2 |
H9 |
106.921 |
| H10 |
C3 |
H11 |
107.359 |
|
Electronic energy levels
Electronic state
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at MP2=FULL/6-31G**
| | hartrees |
|---|
| Energy at 0K | -193.769724 |
| Energy at 298.15K | |
| HF Energy | -193.127007 |
| Nuclear repulsion energy | 133.299859 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2=FULL/6-31G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3901 |
3646 |
23.31 |
101.14 |
0.30 |
0.46 |
| 2 |
A |
3252 |
3039 |
18.21 |
36.48 |
0.74 |
0.85 |
| 3 |
A |
3223 |
3012 |
38.33 |
48.62 |
0.66 |
0.79 |
| 4 |
A |
3192 |
2983 |
19.28 |
71.56 |
0.75 |
0.86 |
| 5 |
A |
3137 |
2931 |
48.52 |
95.27 |
0.07 |
0.13 |
| 6 |
A |
3134 |
2928 |
27.71 |
87.27 |
0.04 |
0.07 |
| 7 |
A |
3120 |
2916 |
36.44 |
75.90 |
0.70 |
0.82 |
| 8 |
A |
3073 |
2871 |
58.80 |
88.49 |
0.12 |
0.21 |
| 9 |
A |
1587 |
1483 |
2.75 |
6.36 |
0.68 |
0.81 |
| 10 |
A |
1573 |
1470 |
6.02 |
9.20 |
0.75 |
0.86 |
| 11 |
A |
1558 |
1456 |
6.10 |
20.07 |
0.75 |
0.86 |
| 12 |
A |
1539 |
1438 |
2.06 |
16.61 |
0.75 |
0.86 |
| 13 |
A |
1499 |
1401 |
7.02 |
2.29 |
0.46 |
0.63 |
| 14 |
A |
1468 |
1372 |
2.72 |
3.02 |
0.75 |
0.85 |
| 15 |
A |
1424 |
1330 |
1.89 |
1.03 |
0.74 |
0.85 |
| 16 |
A |
1364 |
1274 |
14.76 |
15.65 |
0.73 |
0.84 |
| 17 |
A |
1307 |
1221 |
0.84 |
8.29 |
0.75 |
0.85 |
| 18 |
A |
1279 |
1195 |
45.11 |
6.21 |
0.73 |
0.84 |
| 19 |
A |
1201 |
1122 |
7.64 |
1.62 |
0.43 |
0.60 |
| 20 |
A |
1157 |
1081 |
9.17 |
3.80 |
0.54 |
0.70 |
| 21 |
A |
1111 |
1038 |
41.31 |
3.12 |
0.68 |
0.81 |
| 22 |
A |
1020 |
953 |
42.27 |
4.25 |
0.72 |
0.84 |
| 23 |
A |
957 |
894 |
2.66 |
0.35 |
0.72 |
0.83 |
| 24 |
A |
905 |
845 |
1.66 |
9.56 |
0.17 |
0.29 |
| 25 |
A |
797 |
745 |
0.73 |
0.51 |
0.52 |
0.69 |
| 26 |
A |
492 |
460 |
8.29 |
0.24 |
0.53 |
0.69 |
| 27 |
A |
340 |
317 |
11.96 |
0.55 |
0.60 |
0.75 |
| 28 |
A |
272 |
254 |
113.86 |
4.16 |
0.74 |
0.85 |
| 29 |
A |
244 |
228 |
3.87 |
0.07 |
0.68 |
0.81 |
| 30 |
A |
151 |
141 |
8.14 |
0.11 |
0.74 |
0.85 |
Unscaled Zero Point Vibrational Energy (zpe) 24637.6 cm
-1
Scaled (by 0.9344) Zero Point Vibrational Energy (zpe) 23021.3 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2=FULL/6-31G**
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-1.516 |
-0.520 |
0.130 |
| C2 |
-0.633 |
0.646 |
-0.288 |
| C3 |
0.764 |
0.552 |
0.288 |
| O4 |
1.358 |
-0.642 |
-0.216 |
| H5 |
-2.511 |
-0.433 |
-0.302 |
| H6 |
-1.623 |
-0.553 |
1.214 |
| H7 |
-1.079 |
-1.460 |
-0.194 |
| H8 |
-0.550 |
0.683 |
-1.374 |
| H9 |
-1.074 |
1.592 |
0.033 |
| H10 |
0.710 |
0.531 |
1.383 |
| H11 |
1.348 |
1.432 |
0.002 |
| H12 |
2.228 |
-0.729 |
0.186 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
O4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
H12 |
| C1 | | 1.5217 | 2.5246 | 2.8973 | 1.0880 | 1.0894 | 1.0859 | 2.1554 | 2.1596 | 2.7621 | 3.4690 | 3.7508 |
C2 | 1.5217 | | 1.5138 | 2.3716 | 2.1667 | 2.1619 | 2.1547 | 1.0902 | 1.0914 | 2.1465 | 2.1510 | 3.2094 | C3 | 2.5246 | 1.5138 | | 1.4255 | 3.4707 | 2.7883 | 2.7709 | 2.1233 | 2.1266 | 1.0959 | 1.0948 | 1.9482 | O4 | 2.8973 | 2.3716 | 1.4255 | | 3.8753 | 3.3071 | 2.5707 | 2.5952 | 3.3105 | 2.0860 | 2.0854 | 0.9631 | H5 | 1.0880 | 2.1667 | 3.4707 | 3.8753 | | 1.7606 | 1.7649 | 2.4990 | 2.5058 | 3.7607 | 4.2976 | 4.7737 | H6 | 1.0894 | 2.1619 | 2.7883 | 3.3071 | 1.7606 | | 1.7609 | 3.0625 | 2.5089 | 2.5778 | 3.7734 | 3.9900 | H7 | 1.0859 | 2.1547 | 2.7709 | 2.5707 | 1.7649 | 1.7609 | | 2.5031 | 3.0597 | 3.1066 | 3.7810 | 3.4087 | H8 | 2.1554 | 1.0902 | 2.1233 | 2.5952 | 2.4990 | 3.0625 | 2.5031 | | 1.7549 | 3.0353 | 2.4614 | 3.4853 | H9 | 2.1596 | 1.0914 | 2.1266 | 3.3105 | 2.5058 | 2.5089 | 3.0597 | 1.7549 | | 2.4756 | 2.4274 | 4.0386 | H10 | 2.7621 | 2.1465 | 1.0959 | 2.0860 | 3.7607 | 2.5778 | 3.1066 | 3.0353 | 2.4756 | | 1.7685 | 2.3076 | H11 | 3.4690 | 2.1510 | 1.0948 | 2.0854 | 4.2976 | 3.7734 | 3.7810 | 2.4614 | 2.4274 | 1.7685 | | 2.3407 | H12 | 3.7508 | 3.2094 | 1.9482 | 0.9631 | 4.7737 | 3.9900 | 3.4087 | 3.4853 | 4.0386 | 2.3076 | 2.3407 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
112.547 |
|
C1 |
C2 |
H8 |
110.127 |
| C1 |
C2 |
H9 |
110.392 |
|
C2 |
C1 |
H5 |
111.160 |
| C2 |
C1 |
H6 |
110.689 |
|
C2 |
C1 |
H7 |
110.330 |
| C2 |
C3 |
O4 |
107.544 |
|
C2 |
C3 |
H10 |
109.631 |
| C2 |
C3 |
H11 |
110.048 |
|
C3 |
C2 |
H8 |
108.152 |
| C3 |
C2 |
H9 |
108.338 |
|
C3 |
O4 |
H12 |
107.724 |
| O4 |
C3 |
H10 |
110.975 |
|
O4 |
C3 |
H11 |
110.992 |
| H5 |
C1 |
H6 |
107.915 |
|
H5 |
C1 |
H7 |
108.554 |
| H6 |
C1 |
H7 |
108.092 |
|
H8 |
C2 |
H9 |
107.103 |
| H10 |
C3 |
H11 |
107.658 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
