Jump to
S1C2
Energy calculated at MP2/cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -217.746147 |
| Energy at 298.15K | -217.754025 |
| HF Energy | -217.126509 |
| Nuclear repulsion energy | 127.637192 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2/cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3189 |
3037 |
20.51 |
|
|
|
| 2 |
A' |
3101 |
2953 |
26.03 |
|
|
|
| 3 |
A' |
3084 |
2937 |
37.05 |
|
|
|
| 4 |
A' |
3079 |
2933 |
21.00 |
|
|
|
| 5 |
A' |
1523 |
1451 |
1.55 |
|
|
|
| 6 |
A' |
1508 |
1436 |
6.23 |
|
|
|
| 7 |
A' |
1493 |
1422 |
0.19 |
|
|
|
| 8 |
A' |
1446 |
1378 |
17.49 |
|
|
|
| 9 |
A' |
1414 |
1347 |
2.22 |
|
|
|
| 10 |
A' |
1332 |
1269 |
1.73 |
|
|
|
| 11 |
A' |
1153 |
1098 |
1.87 |
|
|
|
| 12 |
A' |
1108 |
1055 |
79.63 |
|
|
|
| 13 |
A' |
1073 |
1022 |
19.13 |
|
|
|
| 14 |
A' |
913 |
870 |
8.14 |
|
|
|
| 15 |
A' |
455 |
433 |
6.05 |
|
|
|
| 16 |
A' |
265 |
253 |
3.14 |
|
|
|
| 17 |
A" |
3180 |
3029 |
56.94 |
|
|
|
| 18 |
A" |
3159 |
3009 |
11.45 |
|
|
|
| 19 |
A" |
3136 |
2987 |
17.19 |
|
|
|
| 20 |
A" |
1500 |
1429 |
6.99 |
|
|
|
| 21 |
A" |
1323 |
1260 |
0.01 |
|
|
|
| 22 |
A" |
1277 |
1217 |
0.66 |
|
|
|
| 23 |
A" |
1201 |
1144 |
1.73 |
|
|
|
| 24 |
A" |
896 |
853 |
1.86 |
|
|
|
| 25 |
A" |
773 |
737 |
1.45 |
|
|
|
| 26 |
A" |
244 |
232 |
0.00 |
|
|
|
| 27 |
A" |
136 |
130 |
2.91 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21480.2 cm
-1
Scaled (by 0.9525) Zero Point Vibrational Energy (zpe) 20459.9 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2/cc-pVDZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.128 |
-0.782 |
0.000 |
| C2 |
0.000 |
0.731 |
0.000 |
| C3 |
-1.466 |
1.169 |
0.000 |
| F4 |
1.469 |
-1.153 |
0.000 |
| H5 |
-0.350 |
-1.216 |
0.896 |
| H6 |
-0.350 |
-1.216 |
-0.896 |
| H7 |
0.523 |
1.128 |
-0.887 |
| H8 |
0.523 |
1.128 |
0.887 |
| H9 |
-1.552 |
2.267 |
0.000 |
| H10 |
-1.995 |
0.791 |
-0.892 |
| H11 |
-1.995 |
0.791 |
0.892 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5178 | 2.5191 | 1.3911 | 1.1050 | 1.1050 | 2.1422 | 2.1422 | 3.4809 | 2.7887 | 2.7887 |
C2 | 1.5178 | | 1.5299 | 2.3886 | 2.1717 | 2.1717 | 1.1039 | 1.1039 | 2.1836 | 2.1861 | 2.1861 | C3 | 2.5191 | 1.5299 | | 3.7421 | 2.7813 | 2.7813 | 2.1780 | 2.1780 | 1.1013 | 1.1036 | 1.1036 | F4 | 1.3911 | 2.3886 | 3.7421 | | 2.0293 | 2.0293 | 2.6237 | 2.6237 | 4.5629 | 4.0710 | 4.0710 | H5 | 1.1050 | 2.1717 | 2.7813 | 2.0293 | | 1.7922 | 3.0721 | 2.5013 | 3.7918 | 3.1509 | 2.5948 | H6 | 1.1050 | 2.1717 | 2.7813 | 2.0293 | 1.7922 | | 2.5013 | 3.0721 | 3.7918 | 2.5948 | 3.1509 | H7 | 2.1422 | 1.1039 | 2.1780 | 2.6237 | 3.0721 | 2.5013 | | 1.7749 | 2.5278 | 2.5404 | 3.1013 | H8 | 2.1422 | 1.1039 | 2.1780 | 2.6237 | 2.5013 | 3.0721 | 1.7749 | | 2.5278 | 3.1013 | 2.5404 | H9 | 3.4809 | 2.1836 | 1.1013 | 4.5629 | 3.7918 | 3.7918 | 2.5278 | 2.5278 | | 1.7804 | 1.7804 | H10 | 2.7887 | 2.1861 | 1.1036 | 4.0710 | 3.1509 | 2.5948 | 2.5404 | 3.1013 | 1.7804 | | 1.7831 | H11 | 2.7887 | 2.1861 | 1.1036 | 4.0710 | 2.5948 | 3.1509 | 3.1013 | 2.5404 | 1.7804 | 1.7831 | |
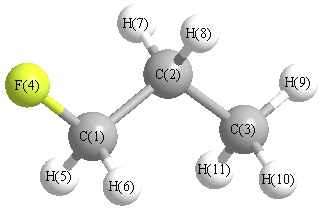 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
111.493 |
|
C1 |
C2 |
H7 |
108.564 |
| C1 |
C2 |
H8 |
108.564 |
|
C2 |
C1 |
F4 |
110.322 |
| C2 |
C1 |
H5 |
110.803 |
|
C2 |
C1 |
H6 |
110.803 |
| C2 |
C3 |
H9 |
111.129 |
|
C2 |
C3 |
H10 |
111.187 |
| C2 |
C3 |
H11 |
111.187 |
|
C3 |
C2 |
H7 |
110.532 |
| C3 |
C2 |
H8 |
110.532 |
|
F4 |
C1 |
H5 |
108.225 |
| F4 |
C1 |
H6 |
108.225 |
|
H5 |
C1 |
H6 |
108.373 |
| H7 |
C2 |
H8 |
107.017 |
|
H9 |
C3 |
H10 |
107.698 |
| H9 |
C3 |
H11 |
107.698 |
|
H10 |
C3 |
H11 |
107.772 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at MP2/cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -217.746981 |
| Energy at 298.15K | |
| HF Energy | -217.127016 |
| Nuclear repulsion energy | 130.181492 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP2/cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3200 |
3048 |
15.92 |
|
|
|
| 2 |
A |
3182 |
3031 |
33.66 |
|
|
|
| 3 |
A |
3155 |
3005 |
44.68 |
|
|
|
| 4 |
A |
3143 |
2994 |
18.50 |
|
|
|
| 5 |
A |
3091 |
2945 |
17.54 |
|
|
|
| 6 |
A |
3088 |
2941 |
30.59 |
|
|
|
| 7 |
A |
3083 |
2937 |
32.88 |
|
|
|
| 8 |
A |
1518 |
1446 |
0.58 |
|
|
|
| 9 |
A |
1509 |
1437 |
7.97 |
|
|
|
| 10 |
A |
1494 |
1423 |
6.52 |
|
|
|
| 11 |
A |
1472 |
1402 |
2.40 |
|
|
|
| 12 |
A |
1439 |
1370 |
15.99 |
|
|
|
| 13 |
A |
1416 |
1349 |
3.05 |
|
|
|
| 14 |
A |
1377 |
1312 |
0.75 |
|
|
|
| 15 |
A |
1308 |
1246 |
1.06 |
|
|
|
| 16 |
A |
1282 |
1221 |
1.30 |
|
|
|
| 17 |
A |
1187 |
1130 |
1.85 |
|
|
|
| 18 |
A |
1137 |
1083 |
13.59 |
|
|
|
| 19 |
A |
1124 |
1071 |
42.44 |
|
|
|
| 20 |
A |
997 |
949 |
37.38 |
|
|
|
| 21 |
A |
926 |
882 |
2.18 |
|
|
|
| 22 |
A |
903 |
860 |
5.08 |
|
|
|
| 23 |
A |
776 |
739 |
0.46 |
|
|
|
| 24 |
A |
483 |
460 |
3.62 |
|
|
|
| 25 |
A |
322 |
307 |
1.02 |
|
|
|
| 26 |
A |
235 |
224 |
2.09 |
|
|
|
| 27 |
A |
150 |
143 |
2.27 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21497.7 cm
-1
Scaled (by 0.9525) Zero Point Vibrational Energy (zpe) 20476.5 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP2/cc-pVDZ
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.800 |
0.516 |
0.294 |
| C2 |
-0.596 |
0.667 |
-0.281 |
| C3 |
-1.514 |
-0.489 |
0.119 |
| F4 |
1.378 |
-0.665 |
-0.172 |
| H5 |
1.453 |
1.355 |
-0.004 |
| H6 |
0.769 |
0.465 |
1.398 |
| H7 |
-0.517 |
0.729 |
-1.381 |
| H8 |
-1.008 |
1.632 |
0.067 |
| H9 |
-2.513 |
-0.385 |
-0.335 |
| H10 |
-1.082 |
-1.447 |
-0.209 |
| H11 |
-1.640 |
-0.531 |
1.215 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5180 | 2.5297 | 1.3948 | 1.1036 | 1.1051 | 2.1413 | 2.1372 | 3.4908 | 2.7660 | 2.8108 |
C2 | 1.5180 | | 1.5295 | 2.3840 | 2.1791 | 2.1733 | 1.1044 | 1.1055 | 2.1870 | 2.1706 | 2.1820 | C3 | 2.5297 | 1.5295 | | 2.9116 | 3.4956 | 2.7859 | 2.1740 | 2.1817 | 1.1016 | 1.1011 | 1.1038 | F4 | 1.3948 | 2.3840 | 2.9116 | | 2.0283 | 2.0272 | 2.6445 | 3.3206 | 3.9038 | 2.5809 | 3.3235 | H5 | 1.1036 | 2.1791 | 3.4956 | 2.0283 | | 1.7956 | 2.4832 | 2.4775 | 4.3432 | 3.7838 | 3.8222 | H6 | 1.1051 | 2.1733 | 2.7859 | 2.0272 | 1.7956 | | 3.0728 | 2.5082 | 3.8074 | 3.1090 | 2.6137 | H7 | 2.1413 | 1.1044 | 2.1740 | 2.6445 | 2.4832 | 3.0728 | | 1.7761 | 2.5137 | 2.5348 | 3.0957 | H8 | 2.1372 | 1.1055 | 2.1817 | 3.3206 | 2.4775 | 2.5082 | 1.7761 | | 2.5488 | 3.0928 | 2.5290 | H9 | 3.4908 | 2.1870 | 1.1016 | 3.9038 | 4.3432 | 3.8074 | 2.5137 | 2.5488 | | 1.7865 | 1.7841 | H10 | 2.7660 | 2.1706 | 1.1011 | 2.5809 | 3.7838 | 3.1090 | 2.5348 | 3.0928 | 1.7865 | | 1.7829 | H11 | 2.8108 | 2.1820 | 1.1038 | 3.3235 | 3.8222 | 2.6137 | 3.0957 | 2.5290 | 1.7841 | 1.7829 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
112.212 |
|
C1 |
C2 |
H7 |
108.445 |
| C1 |
C2 |
H8 |
108.072 |
|
C2 |
C1 |
F4 |
109.786 |
| C2 |
C1 |
H5 |
111.468 |
|
C2 |
C1 |
H6 |
110.910 |
| C2 |
C3 |
H9 |
111.408 |
|
C2 |
C3 |
H10 |
110.134 |
| C2 |
C3 |
H11 |
110.883 |
|
C3 |
C2 |
H7 |
110.209 |
| C3 |
C2 |
H8 |
110.752 |
|
F4 |
C1 |
H5 |
107.982 |
| F4 |
C1 |
H6 |
107.807 |
|
H5 |
C1 |
H6 |
108.770 |
| H7 |
C2 |
H8 |
106.971 |
|
H9 |
C3 |
H10 |
108.396 |
| H9 |
C3 |
H11 |
107.989 |
|
H10 |
C3 |
H11 |
107.917 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
