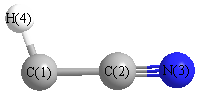Jump to
S1C2
S2C1
S2C2
Energy calculated at MP4=FULL/aug-cc-pVTZ
| | hartrees |
|---|
| Energy at 0K | -131.235761 |
| Energy at 298.15K | -131.235261 |
| HF Energy | -130.723689 |
| Nuclear repulsion energy | 47.747672 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP4=FULL/aug-cc-pVTZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3381 |
3277 |
|
|
|
|
| 2 |
A' |
2037 |
1975 |
|
|
|
|
| 3 |
A' |
1138 |
1103 |
|
|
|
|
| 4 |
A' |
650 |
630 |
|
|
|
|
| 5 |
A' |
417 |
404 |
|
|
|
|
| 6 |
A" |
501 |
486 |
|
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 4061.8 cm
-1
Scaled (by 0.9693) Zero Point Vibrational Energy (zpe) 3937.1 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP4=FULL/aug-cc-pVTZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-0.114 |
-1.227 |
0.000 |
| C2 |
0.000 |
0.105 |
0.000 |
| N3 |
0.033 |
1.266 |
0.000 |
| H4 |
0.455 |
-2.127 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
N3 |
H4 |
| C1 | | 1.3372 | 2.4973 | 1.0648 |
C2 | 1.3372 | | 1.1611 | 2.2782 | N3 | 2.4973 | 1.1611 | | 3.4190 | H4 | 1.0648 | 2.2782 | 3.4190 | |
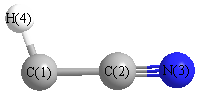 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
N3 |
176.724 |
|
C2 |
C1 |
H4 |
142.791 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
S2C1
S2C2
Energy calculated at MP4=FULL/aug-cc-pVTZ
| | hartrees |
|---|
| Energy at 0K | -131.233742 |
| Energy at 298.15K | |
| HF Energy | -130.721968 |
| Nuclear repulsion energy | 47.910335 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP4=FULL/aug-cc-pVTZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
Σ |
3460 |
3354 |
|
|
|
|
| 2 |
Σ |
1692 |
1640 |
|
|
|
|
| 3 |
Σ |
1250 |
1212 |
|
|
|
|
| 4 |
Π |
502 |
487 |
|
|
|
|
| 4 |
Π |
502 |
487 |
|
|
|
|
| 5 |
Π |
360i |
349i |
|
|
|
|
| 5 |
Π |
360i |
349i |
|
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 3342.9 cm
-1
Scaled (by 0.9693) Zero Point Vibrational Energy (zpe) 3240.3 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP4=FULL/aug-cc-pVTZ
Point Group is C∞v
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.000 |
0.000 |
-1.202 |
| C2 |
0.000 |
0.000 |
0.092 |
| N3 |
0.000 |
0.000 |
1.274 |
| H4 |
0.000 |
0.000 |
-2.259 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
N3 |
H4 |
| C1 | | 1.2933 | 2.4754 | 1.0573 |
C2 | 1.2933 | | 1.1822 | 2.3506 | N3 | 2.4754 | 1.1822 | | 3.5328 | H4 | 1.0573 | 2.3506 | 3.5328 | |
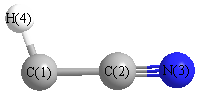 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
N3 |
180.000 |
|
C2 |
C1 |
H4 |
180.000 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at MP4=FULL/aug-cc-pVTZ
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
-0.815 |
|
|
|
| 2 |
C |
0.771 |
|
|
|
| 3 |
N |
-0.509 |
|
|
|
| 4 |
H |
0.553 |
|
|
|
Electric dipole moments
Electric Quadrupole moment
Quadrupole components in D Å
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
0.000 |
0.000 |
0.000 |
| y |
0.000 |
0.000 |
0.000 |
| z |
0.000 |
0.000 |
0.000 |
<r2> (average value of r
2) Å
2
| <r2> |
35.710 |
| (<r2>)1/2 |
5.976 |
Jump to
S1C1
S1C2
S2C2
Energy calculated at MP4=FULL/aug-cc-pVTZ
| | hartrees |
|---|
| Energy at 0K | -131.230402 |
| Energy at 298.15K | -131.229822 |
| HF Energy | -130.662080 |
| Nuclear repulsion energy | 46.837235 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP4=FULL/aug-cc-pVTZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3084 |
2989 |
|
|
|
|
| 2 |
A' |
2082 |
2018 |
|
|
|
|
| 3 |
A' |
1060 |
1028 |
|
|
|
|
| 4 |
A' |
972 |
942 |
|
|
|
|
| 5 |
A' |
425 |
412 |
|
|
|
|
| 6 |
A" |
309 |
300 |
|
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 3966.1 cm
-1
Scaled (by 0.9693) Zero Point Vibrational Energy (zpe) 3844.4 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP4=FULL/aug-cc-pVTZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.098 |
-1.288 |
0.000 |
| C2 |
0.000 |
0.093 |
0.000 |
| N3 |
-0.249 |
1.251 |
0.000 |
| H4 |
1.152 |
-1.581 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
N3 |
H4 |
| C1 | | 1.3848 | 2.5626 | 1.0936 |
C2 | 1.3848 | | 1.1841 | 2.0319 | N3 | 2.5626 | 1.1841 | | 3.1590 | H4 | 1.0936 | 2.0319 | 3.1590 | |
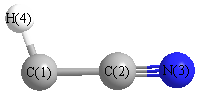 More geometry information
More geometry information
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
S1C2
S2C1
Energy calculated at MP4=FULL/aug-cc-pVTZ
| | hartrees |
|---|
| Energy at 0K | -131.230402 |
| Energy at 298.15K | -131.229822 |
| HF Energy | -130.662080 |
| Nuclear repulsion energy | 46.837235 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP4=FULL/aug-cc-pVTZ
Geometric Data calculated at MP4=FULL/aug-cc-pVTZ
Point Group is Cs
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability