Jump to
S1C2
Energy calculated at PBE1PBE/aug-cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -958.712609 |
| Energy at 298.15K | |
| HF Energy | -958.712609 |
| Nuclear repulsion energy | 125.790341 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at PBE1PBE/aug-cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A1 |
3284 |
3158 |
0.99 |
|
|
|
| 2 |
A1 |
772 |
742 |
6.84 |
|
|
|
| 3 |
A1 |
312 |
300 |
0.38 |
|
|
|
| 4 |
B1 |
364i |
350i |
35.63 |
|
|
|
| 5 |
B2 |
1217 |
1171 |
53.48 |
|
|
|
| 6 |
B2 |
947 |
910 |
139.87 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 3083.8 cm
-1
Scaled (by 0.9616) Zero Point Vibrational Energy (zpe) 2965.4 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at PBE1PBE/aug-cc-pVDZ
Point Group is C2v
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.000 |
0.000 |
0.675 |
| H2 |
0.000 |
0.000 |
1.760 |
| Cl3 |
0.000 |
1.473 |
-0.171 |
| Cl4 |
0.000 |
-1.473 |
-0.171 |
Atom - Atom Distances (Å)
| |
C1 |
H2 |
Cl3 |
Cl4 |
| C1 | | 1.0852 | 1.6987 | 1.6987 |
H2 | 1.0852 | | 2.4289 | 2.4289 | Cl3 | 1.6987 | 2.4289 | | 2.9462 | Cl4 | 1.6987 | 2.4289 | 2.9462 | |
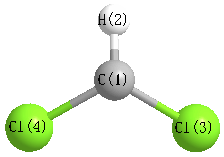 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| Cl3 |
C1 |
H2 |
119.866 |
|
Cl3 |
C1 |
Cl4 |
120.268 |
| Cl4 |
C1 |
H2 |
119.866 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at PBE1PBE/aug-cc-pVDZ
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
0.002 |
|
|
|
| 2 |
H |
-0.077 |
|
|
|
| 3 |
Cl |
0.037 |
|
|
|
| 4 |
Cl |
0.037 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
0.000 |
0.000 |
0.953 |
0.953 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-32.162 |
0.000 |
0.000 |
| y |
0.000 |
-31.016 |
0.000 |
| z |
0.000 |
0.000 |
-28.999 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
-2.154 |
0.000 |
0.000 |
| y |
0.000 |
-0.435 |
0.000 |
| z |
0.000 |
0.000 |
2.590 |
|
| Polar |
|---|
| 3z2-r2 | 5.179 |
|---|
| x2-y2 | -1.146 |
|---|
| xy | 0.000 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
4.538 |
0.000 |
0.000 |
| y |
0.000 |
8.094 |
0.000 |
| z |
0.000 |
0.000 |
5.377 |
<r2> (average value of r
2) Å
2
| <r2> |
99.798 |
| (<r2>)1/2 |
9.990 |
Jump to
S1C1
Energy calculated at PBE1PBE/aug-cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -958.713442 |
| Energy at 298.15K | -958.714251 |
| HF Energy | -958.713442 |
| Nuclear repulsion energy | 125.649859 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at PBE1PBE/aug-cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3239 |
3114 |
0.66 |
|
|
|
| 2 |
A' |
776 |
746 |
11.41 |
|
|
|
| 3 |
A' |
484 |
466 |
21.94 |
|
|
|
| 4 |
A' |
305 |
293 |
0.58 |
|
|
|
| 5 |
A" |
1219 |
1172 |
41.65 |
|
|
|
| 6 |
A" |
914 |
879 |
166.14 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 3468.1 cm
-1
Scaled (by 0.9616) Zero Point Vibrational Energy (zpe) 3334.9 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at PBE1PBE/aug-cc-pVDZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.012 |
0.700 |
0.000 |
| H2 |
-0.497 |
1.662 |
0.000 |
| Cl3 |
0.012 |
-0.172 |
1.469 |
| Cl4 |
0.012 |
-0.172 |
-1.469 |
Atom - Atom Distances (Å)
| |
C1 |
H2 |
Cl3 |
Cl4 |
| C1 | | 1.0888 | 1.7083 | 1.7083 |
H2 | 1.0888 | | 2.4047 | 2.4047 | Cl3 | 1.7083 | 2.4047 | | 2.9377 | Cl4 | 1.7083 | 2.4047 | 2.9377 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| Cl3 |
C1 |
H2 |
116.831 |
|
Cl3 |
C1 |
Cl4 |
118.597 |
| Cl4 |
C1 |
H2 |
116.831 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at PBE1PBE/aug-cc-pVDZ
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
-0.077 |
|
|
|
| 2 |
H |
-0.016 |
|
|
|
| 3 |
Cl |
0.047 |
|
|
|
| 4 |
Cl |
0.047 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
-0.450 |
0.861 |
0.000 |
0.972 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-31.856 |
-0.935 |
0.000 |
| y |
-0.935 |
-29.445 |
0.000 |
| z |
0.000 |
0.000 |
-31.120 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
-1.574 |
-0.935 |
0.000 |
| y |
-0.935 |
2.043 |
0.000 |
| z |
0.000 |
0.000 |
-0.469 |
|
| Polar |
|---|
| 3z2-r2 | -0.938 |
|---|
| x2-y2 | -2.411 |
|---|
| xy | -0.935 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
4.582 |
-0.065 |
0.000 |
| y |
-0.065 |
5.411 |
0.000 |
| z |
0.000 |
0.000 |
8.181 |
<r2> (average value of r
2) Å
2
| <r2> |
99.562 |
| (<r2>)1/2 |
9.978 |
