Jump to
S1C2
Energy calculated at HSEh1PBE/6-311+G(3df,2p)
| | hartrees |
|---|
| Energy at 0K | -188.983553 |
| Energy at 298.15K | -188.984656 |
| HF Energy | -188.983553 |
| Nuclear repulsion energy | 63.613397 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at HSEh1PBE/6-311+G(3df,2p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3653 |
3653 |
21.99 |
|
|
|
| 2 |
A' |
1913 |
1913 |
357.92 |
|
|
|
| 3 |
A' |
1303 |
1303 |
0.09 |
|
|
|
| 4 |
A' |
1107 |
1107 |
164.74 |
|
|
|
| 5 |
A' |
606 |
606 |
34.14 |
|
|
|
| 6 |
A" |
599 |
599 |
108.19 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 4590.3 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 4590.3 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at HSEh1PBE/6-311+G(3df,2p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.000 |
0.435 |
0.000 |
| O2 |
-1.055 |
-0.351 |
0.000 |
| O3 |
1.151 |
0.185 |
0.000 |
| H4 |
-0.768 |
-1.280 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
O3 |
H4 |
| C1 | | 1.3148 | 1.1773 | 1.8790 |
O2 | 1.3148 | | 2.2691 | 0.9729 | O3 | 1.1773 | 2.2691 | | 2.4137 | H4 | 1.8790 | 0.9729 | 2.4137 | |
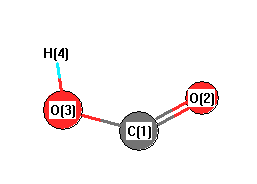 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
O2 |
H4 |
109.531 |
|
O2 |
C1 |
O3 |
131.067 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at HSEh1PBE/6-311+G(3df,2p)
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
0.654 |
|
|
|
| 2 |
O |
-0.381 |
|
|
|
| 3 |
O |
-0.495 |
|
|
|
| 4 |
H |
0.221 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
-1.110 |
-1.670 |
0.000 |
2.005 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-20.225 |
0.796 |
0.000 |
| y |
0.796 |
-14.076 |
0.000 |
| z |
0.000 |
0.000 |
-16.370 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
-5.002 |
0.796 |
0.000 |
| y |
0.796 |
4.222 |
0.000 |
| z |
0.000 |
0.000 |
0.780 |
|
| Polar |
|---|
| 3z2-r2 | 1.560 |
|---|
| x2-y2 | -6.149 |
|---|
| xy | 0.796 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
3.896 |
0.186 |
0.000 |
| y |
0.186 |
3.408 |
0.000 |
| z |
0.000 |
0.000 |
2.346 |
<r2> (average value of r
2) Å
2
| <r2> |
34.654 |
| (<r2>)1/2 |
5.887 |
Jump to
S1C1
Energy calculated at HSEh1PBE/6-311+G(3df,2p)
| | hartrees |
|---|
| Energy at 0K | -188.986248 |
| Energy at 298.15K | -188.987336 |
| HF Energy | -188.986248 |
| Nuclear repulsion energy | 63.373821 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at HSEh1PBE/6-311+G(3df,2p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3863 |
3863 |
133.42 |
|
|
|
| 2 |
A' |
1948 |
1948 |
265.89 |
|
|
|
| 3 |
A' |
1253 |
1253 |
266.56 |
|
|
|
| 4 |
A' |
1123 |
1123 |
47.89 |
|
|
|
| 5 |
A' |
630 |
630 |
4.38 |
|
|
|
| 6 |
A" |
555 |
555 |
85.33 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 4685.7 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 4685.7 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at HSEh1PBE/6-311+G(3df,2p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.000 |
0.403 |
0.000 |
| O2 |
-0.938 |
-0.540 |
0.000 |
| O3 |
1.163 |
0.252 |
0.000 |
| H4 |
-1.802 |
-0.117 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
O3 |
H4 |
| C1 | | 1.3292 | 1.1725 | 1.8752 |
O2 | 1.3292 | | 2.2446 | 0.9619 | O3 | 1.1725 | 2.2446 | | 2.9874 | H4 | 1.8752 | 0.9619 | 2.9874 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
O2 |
H4 |
108.813 |
|
O2 |
C1 |
O3 |
127.487 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at HSEh1PBE/6-311+G(3df,2p)
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
0.609 |
|
|
|
| 2 |
O |
-0.385 |
|
|
|
| 3 |
O |
-0.477 |
|
|
|
| 4 |
H |
0.253 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
-3.029 |
0.295 |
0.000 |
3.043 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-14.081 |
-0.936 |
0.000 |
| y |
-0.936 |
-18.119 |
0.000 |
| z |
0.000 |
0.000 |
-16.424 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
3.191 |
-0.936 |
0.000 |
| y |
-0.936 |
-2.867 |
0.000 |
| z |
0.000 |
0.000 |
-0.323 |
|
| Polar |
|---|
| 3z2-r2 | -0.647 |
|---|
| x2-y2 | 4.039 |
|---|
| xy | -0.936 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
4.277 |
0.124 |
0.000 |
| y |
0.124 |
2.977 |
0.000 |
| z |
0.000 |
0.000 |
2.400 |
<r2> (average value of r
2) Å
2
| <r2> |
35.042 |
| (<r2>)1/2 |
5.920 |
