Jump to
S1C2
Energy calculated at HSEh1PBE/6-31+G**
| | hartrees |
|---|
| Energy at 0K | -499.248961 |
| Energy at 298.15K | |
| HF Energy | -499.248961 |
| Nuclear repulsion energy | 45.388089 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at HSEh1PBE/6-31+G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3211 |
3066 |
5.96 |
108.24 |
0.11 |
0.20 |
| 2 |
A' |
1429 |
1364 |
11.23 |
2.21 |
0.70 |
0.83 |
| 3 |
A' |
865 |
826 |
40.01 |
6.78 |
0.22 |
0.36 |
| 4 |
A' |
231 |
220 |
102.94 |
0.15 |
0.75 |
0.86 |
| 5 |
A" |
3366 |
3214 |
0.08 |
50.43 |
0.75 |
0.86 |
| 6 |
A" |
1013 |
967 |
0.61 |
3.74 |
0.75 |
0.86 |
Unscaled Zero Point Vibrational Energy (zpe) 5057.1 cm
-1
Scaled (by 0.9548) Zero Point Vibrational Energy (zpe) 4828.5 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at HSEh1PBE/6-31+G**
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-0.000 |
1.116 |
0.000 |
| Cl2 |
-0.000 |
-0.584 |
0.000 |
| H3 |
0.000 |
1.620 |
0.956 |
| H4 |
0.000 |
1.620 |
-0.956 |
Atom - Atom Distances (Å)
| |
C1 |
Cl2 |
H3 |
H4 |
| C1 | | 1.7003 | 1.0803 | 1.0803 |
Cl2 | 1.7003 | | 2.4026 | 2.4026 | H3 | 1.0803 | 2.4026 | | 1.9110 | H4 | 1.0803 | 2.4026 | 1.9110 | |
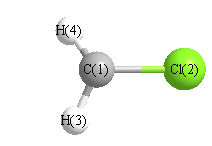 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| Br2 |
C1 |
H3 |
117.812 |
|
Br2 |
C1 |
H4 |
117.812 |
| H3 |
C1 |
H4 |
124.375 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at HSEh1PBE/6-31+G**
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
-0.461 |
|
|
|
| 2 |
Cl |
0.054 |
|
|
|
| 3 |
H |
0.203 |
|
|
|
| 4 |
H |
0.203 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
0.000 |
1.179 |
0.000 |
1.179 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-20.737 |
0.000 |
0.000 |
| y |
0.000 |
-17.450 |
0.000 |
| z |
0.000 |
0.000 |
-18.492 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
-2.766 |
0.000 |
0.000 |
| y |
0.000 |
2.164 |
0.000 |
| z |
0.000 |
0.000 |
0.602 |
|
| Polar |
|---|
| 3z2-r2 | 1.204 |
|---|
| x2-y2 | -3.287 |
|---|
| xy | 0.000 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
2.412 |
0.000 |
0.000 |
| y |
0.000 |
4.621 |
0.000 |
| z |
0.000 |
0.000 |
2.674 |
<r2> (average value of r
2) Å
2
| <r2> |
32.153 |
| (<r2>)1/2 |
5.670 |
Jump to
S1C1
Energy calculated at HSEh1PBE/6-31+G**
| | hartrees |
|---|
| Energy at 0K | -499.248961 |
| Energy at 298.15K | |
| HF Energy | -499.248961 |
| Nuclear repulsion energy | 45.397455 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at HSEh1PBE/6-31+G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A1 |
3211 |
3066 |
6.00 |
108.34 |
0.11 |
0.20 |
| 2 |
A1 |
1429 |
1365 |
11.22 |
|
|
|
| 3 |
A1 |
866 |
827 |
39.92 |
|
|
|
| 4 |
B1 |
234 |
224 |
102.88 |
|
|
|
| 5 |
B2 |
3365 |
3213 |
0.07 |
|
|
|
| 6 |
B2 |
1013 |
967 |
0.62 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 5059.2 cm
-1
Scaled (by 0.9548) Zero Point Vibrational Energy (zpe) 4830.5 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at HSEh1PBE/6-31+G**
Point Group is C2v
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.000 |
0.000 |
-1.115 |
| Cl2 |
0.000 |
0.000 |
0.584 |
| H3 |
0.000 |
0.955 |
-1.620 |
| H4 |
0.000 |
-0.955 |
-1.620 |
Atom - Atom Distances (Å)
| |
C1 |
Cl2 |
H3 |
H4 |
| C1 | | 1.6998 | 1.0804 | 1.0804 |
Cl2 | 1.6998 | | 2.4027 | 2.4027 | H3 | 1.0804 | 2.4027 | | 1.9103 | H4 | 1.0804 | 2.4027 | 1.9103 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| Br2 |
C1 |
H3 |
117.861 |
|
Br2 |
C1 |
H4 |
117.861 |
| H3 |
C1 |
H4 |
124.278 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at HSEh1PBE/6-31+G**
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
-0.461 |
|
|
|
| 2 |
Cl |
0.054 |
|
|
|
| 3 |
H |
0.203 |
|
|
|
| 4 |
H |
0.203 |
|
|
|
Electric dipole moments
Electric dipole components in Debye
(What's a Debye? See section
VII.A.3)
| |
x |
y |
z |
Total |
| |
0.000 |
0.000 |
-1.177 |
1.177 |
| CHELPG |
|
|
|
|
| AIM |
|
|
|
|
| ESP |
|
|
|
|
Electric Quadrupole moment
Quadrupole components in D Å
| Primitive |
|---|
| | x | y | z |
|---|
| x |
-20.736 |
0.000 |
0.000 |
| y |
0.000 |
-18.494 |
0.000 |
| z |
0.000 |
0.000 |
-17.449 |
|
| Traceless |
|---|
| | x | y | z |
|---|
| x |
-2.765 |
0.000 |
0.000 |
| y |
0.000 |
0.598 |
0.000 |
| z |
0.000 |
0.000 |
2.167 |
|
| Polar |
|---|
| 3z2-r2 | 4.333 |
|---|
| x2-y2 | -2.242 |
|---|
| xy | 0.000 |
|---|
| xz | 0.000 |
|---|
| yz | 0.000 |
|---|
|
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
2.412 |
0.000 |
0.000 |
| y |
0.000 |
2.673 |
0.000 |
| z |
0.000 |
0.000 |
4.621 |
<r2> (average value of r
2) Å
2
| <r2> |
32.146 |
| (<r2>)1/2 |
5.670 |
