Jump to
S1C2
Vibrational Frequencies calculated at CCD/6-311G**
Geometric Data calculated at CCD/6-311G**
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at CCD/6-311G**
| | hartrees |
|---|
| Energy at 0K | -230.612718 |
| Energy at 298.15K | -230.618809 |
| HF Energy | -229.820217 |
| Nuclear repulsion energy | 161.720076 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at CCD/6-311G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3297 |
3149 |
11.23 |
|
|
|
| 2 |
A |
3296 |
3148 |
3.11 |
|
|
|
| 3 |
A |
3237 |
3092 |
11.32 |
|
|
|
| 4 |
A |
3205 |
3061 |
5.79 |
|
|
|
| 5 |
A |
3200 |
3057 |
1.27 |
|
|
|
| 6 |
A |
3186 |
3043 |
6.05 |
|
|
|
| 7 |
A |
1741 |
1662 |
87.91 |
|
|
|
| 8 |
A |
1724 |
1646 |
136.01 |
|
|
|
| 9 |
A |
1461 |
1396 |
1.88 |
|
|
|
| 10 |
A |
1444 |
1379 |
25.25 |
|
|
|
| 11 |
A |
1380 |
1318 |
51.00 |
|
|
|
| 12 |
A |
1345 |
1284 |
3.54 |
|
|
|
| 13 |
A |
1269 |
1212 |
246.36 |
|
|
|
| 14 |
A |
1155 |
1103 |
15.25 |
|
|
|
| 15 |
A |
1024 |
978 |
36.16 |
|
|
|
| 16 |
A |
1004 |
959 |
10.17 |
|
|
|
| 17 |
A |
993 |
948 |
39.49 |
|
|
|
| 18 |
A |
902 |
862 |
18.24 |
|
|
|
| 19 |
A |
875 |
836 |
43.02 |
|
|
|
| 20 |
A |
856 |
818 |
49.90 |
|
|
|
| 21 |
A |
748 |
715 |
5.54 |
|
|
|
| 22 |
A |
725 |
692 |
1.22 |
|
|
|
| 23 |
A |
588 |
562 |
2.75 |
|
|
|
| 24 |
A |
464 |
443 |
4.02 |
|
|
|
| 25 |
A |
297 |
283 |
1.62 |
|
|
|
| 26 |
A |
194 |
185 |
8.20 |
|
|
|
| 27 |
A |
94 |
90 |
0.76 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 19850.3 cm
-1
Scaled (by 0.9551) Zero Point Vibrational Energy (zpe) 18959.0 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at CCD/6-311G**
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| O1 |
-0.037 |
-0.831 |
0.051 |
| C2 |
1.283 |
-0.483 |
-0.017 |
| C3 |
-0.921 |
0.175 |
0.355 |
| C4 |
1.785 |
0.748 |
-0.147 |
| C5 |
-2.116 |
0.246 |
-0.229 |
| H6 |
1.907 |
-1.369 |
0.034 |
| H7 |
-0.591 |
0.853 |
1.139 |
| H8 |
2.861 |
0.871 |
-0.182 |
| H9 |
1.165 |
1.632 |
-0.242 |
| H10 |
-2.828 |
0.999 |
0.086 |
| H11 |
-2.398 |
-0.452 |
-1.010 |
Atom - Atom Distances (Å)
| |
O1 |
C2 |
C3 |
C4 |
C5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| O1 | | 1.3667 | 1.3733 | 2.4196 | 2.3580 | 2.0175 | 2.0803 | 3.3691 | 2.7569 | 3.3378 | 2.6156 |
C2 | 1.3667 | | 2.3299 | 1.3361 | 3.4825 | 1.0853 | 2.5750 | 2.0859 | 2.1303 | 4.3711 | 3.8123 | C3 | 1.3733 | 2.3299 | | 2.8116 | 1.3317 | 3.2383 | 1.0886 | 3.8830 | 2.6142 | 2.0947 | 2.1060 | C4 | 2.4196 | 1.3361 | 2.8116 | | 3.9343 | 2.1286 | 2.7035 | 1.0834 | 1.0837 | 4.6259 | 4.4366 | C5 | 2.3580 | 3.4825 | 1.3317 | 3.9343 | | 4.3433 | 2.1375 | 5.0163 | 3.5624 | 1.0836 | 1.0845 | H6 | 2.0175 | 1.0853 | 3.2383 | 2.1286 | 4.3433 | | 3.5210 | 2.4442 | 3.1039 | 5.2948 | 4.5238 | H7 | 2.0803 | 2.5750 | 1.0886 | 2.7035 | 2.1375 | 3.5210 | | 3.6958 | 2.3660 | 2.4774 | 3.0967 | H8 | 3.3691 | 2.0859 | 3.8830 | 1.0834 | 5.0163 | 2.4442 | 3.6958 | | 1.8596 | 5.6968 | 5.4856 | H9 | 2.7569 | 2.1303 | 2.6142 | 1.0837 | 3.5624 | 3.1039 | 2.3660 | 1.8596 | | 4.0566 | 4.1991 | H10 | 3.3378 | 4.3711 | 2.0947 | 4.6259 | 1.0836 | 5.2948 | 2.4774 | 5.6968 | 4.0566 | | 1.8690 | H11 | 2.6156 | 3.8123 | 2.1060 | 4.4366 | 1.0845 | 4.5238 | 3.0967 | 5.4856 | 4.1991 | 1.8690 | |
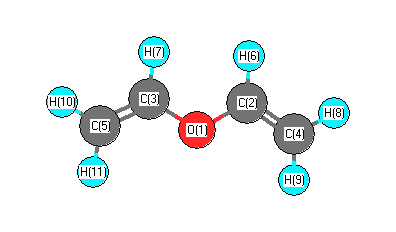 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| O1 |
C2 |
C4 |
127.069 |
|
O1 |
C2 |
H6 |
110.201 |
| O1 |
C3 |
C5 |
121.310 |
|
O1 |
C3 |
H7 |
114.847 |
| C2 |
O1 |
C3 |
116.488 |
|
C2 |
C4 |
H8 |
118.746 |
| C2 |
C4 |
H9 |
123.033 |
|
C3 |
C5 |
H10 |
119.934 |
| C3 |
C5 |
H11 |
120.957 |
|
C4 |
C2 |
H6 |
122.723 |
| C5 |
C3 |
H7 |
123.739 |
|
H8 |
C4 |
H9 |
118.206 |
| H10 |
C5 |
H11 |
119.101 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
