All results from a given calculation for C4H9SH (1-Butanethiol)
using model chemistry: MP3/6-311+G(3df,2p)
19 10 17 12 22
States and conformations
| State |
Conformation |
minimum conformation |
conformer description |
state description |
| 1 |
1 |
yes |
CS |
1A' |
Energy calculated at MP3/6-311+G(3df,2p)
| | hartrees |
|---|
| Energy at 0K | -555.828437 |
| Energy at 298.15K | |
| HF Energy | -554.892711 |
| Nuclear repulsion energy | 223.885952 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/6-311+G(3df,2p)
Geometric Data calculated at MP3/6-311+G(3df,2p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| S1 |
1.390 |
-1.832 |
0.000 |
| C2 |
-0.224 |
-0.988 |
0.000 |
| C3 |
0.000 |
0.520 |
0.000 |
| C4 |
-1.318 |
1.294 |
0.000 |
| C5 |
-1.099 |
2.805 |
0.000 |
| H6 |
0.925 |
-3.085 |
0.000 |
| H7 |
-0.785 |
-1.285 |
0.885 |
| H8 |
-0.785 |
-1.285 |
-0.885 |
| H9 |
0.589 |
0.799 |
-0.877 |
| H10 |
0.589 |
0.799 |
0.877 |
| H11 |
-1.905 |
1.006 |
0.876 |
| H12 |
-1.905 |
1.006 |
-0.876 |
| H13 |
-2.046 |
3.343 |
0.000 |
| H14 |
-0.536 |
3.116 |
0.881 |
| H15 |
-0.536 |
3.116 |
-0.881 |
Atom - Atom Distances (Å)
| |
S1 |
C2 |
C3 |
C4 |
C5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
H12 |
H13 |
H14 |
H15 |
| S1 | | 1.8217 | 2.7321 | 4.1353 | 5.2625 | 1.3363 | 2.4111 | 2.4111 | 2.8870 | 2.8870 | 4.4361 | 4.4361 | 6.2118 | 5.3826 | 5.3826 |
C2 | 1.8217 | | 1.5250 | 2.5302 | 3.8926 | 2.3908 | 1.0885 | 1.0885 | 2.1510 | 2.1510 | 2.7512 | 2.7512 | 4.6988 | 4.2097 | 4.2097 | C3 | 2.7321 | 1.5250 | | 1.5279 | 2.5353 | 3.7215 | 2.1578 | 2.1578 | 1.0930 | 1.0930 | 2.1521 | 2.1521 | 3.4864 | 2.7936 | 2.7936 | C4 | 4.1353 | 2.5302 | 1.5279 | | 1.5269 | 4.9192 | 2.7774 | 2.7774 | 2.1562 | 2.1562 | 1.0928 | 1.0928 | 2.1751 | 2.1699 | 2.1699 | C5 | 5.2625 | 3.8926 | 2.5353 | 1.5269 | | 6.2275 | 4.1958 | 4.1958 | 2.7642 | 2.7642 | 2.1564 | 2.1564 | 1.0891 | 1.0905 | 1.0905 | H6 | 1.3363 | 2.3908 | 3.7215 | 4.9192 | 6.2275 | | 2.6357 | 2.6357 | 3.9959 | 3.9959 | 5.0509 | 5.0509 | 7.0811 | 6.4315 | 6.4315 | H7 | 2.4111 | 1.0885 | 2.1578 | 2.7774 | 4.1958 | 2.6357 | | 1.7696 | 3.0555 | 2.4961 | 2.5501 | 3.0988 | 4.8774 | 4.4082 | 4.7485 | H8 | 2.4111 | 1.0885 | 2.1578 | 2.7774 | 4.1958 | 2.6357 | 1.7696 | | 2.4961 | 3.0555 | 3.0988 | 2.5501 | 4.8774 | 4.7485 | 4.4082 | H9 | 2.8870 | 2.1510 | 1.0930 | 2.1562 | 2.7642 | 3.9959 | 3.0555 | 2.4961 | | 1.7547 | 3.0553 | 2.5023 | 3.7660 | 3.1187 | 2.5760 | H10 | 2.8870 | 2.1510 | 1.0930 | 2.1562 | 2.7642 | 3.9959 | 2.4961 | 3.0555 | 1.7547 | | 2.5023 | 3.0553 | 3.7660 | 2.5760 | 3.1187 | H11 | 4.4361 | 2.7512 | 2.1521 | 1.0928 | 2.1564 | 5.0509 | 2.5501 | 3.0988 | 3.0553 | 2.5023 | | 1.7516 | 2.4994 | 2.5149 | 3.0675 | H12 | 4.4361 | 2.7512 | 2.1521 | 1.0928 | 2.1564 | 5.0509 | 3.0988 | 2.5501 | 2.5023 | 3.0553 | 1.7516 | | 2.4994 | 3.0675 | 2.5149 | H13 | 6.2118 | 4.6988 | 3.4864 | 2.1751 | 1.0891 | 7.0811 | 4.8774 | 4.8774 | 3.7660 | 3.7660 | 2.4994 | 2.4994 | | 1.7621 | 1.7621 | H14 | 5.3826 | 4.2097 | 2.7936 | 2.1699 | 1.0905 | 6.4315 | 4.4082 | 4.7485 | 3.1187 | 2.5760 | 2.5149 | 3.0675 | 1.7621 | | 1.7611 | H15 | 5.3826 | 4.2097 | 2.7936 | 2.1699 | 1.0905 | 6.4315 | 4.7485 | 4.4082 | 2.5760 | 3.1187 | 3.0675 | 2.5149 | 1.7621 | 1.7611 | |
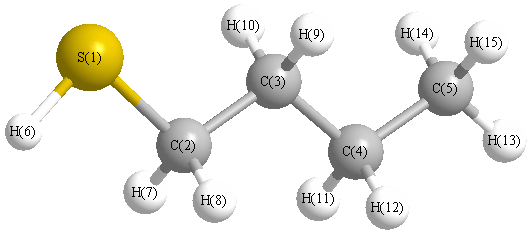 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| S1 |
C2 |
C3 |
109.122 |
|
S1 |
C2 |
H7 |
109.286 |
| S1 |
C2 |
H8 |
109.286 |
|
C2 |
S1 |
H6 |
97.219 |
| C2 |
C3 |
C4 |
111.952 |
|
C2 |
C3 |
H9 |
109.389 |
| C2 |
C3 |
H10 |
109.389 |
|
C3 |
C2 |
H7 |
110.190 |
| C3 |
C2 |
H8 |
110.190 |
|
C3 |
C4 |
C5 |
112.187 |
| C3 |
C4 |
H11 |
109.286 |
|
C3 |
C4 |
H12 |
109.286 |
| C4 |
C3 |
H9 |
109.595 |
|
C4 |
C3 |
H10 |
109.595 |
| C4 |
C5 |
H13 |
111.399 |
|
C4 |
C5 |
H14 |
110.902 |
| C4 |
C5 |
H15 |
110.902 |
|
C5 |
C4 |
H11 |
109.694 |
| C5 |
C4 |
H12 |
109.694 |
|
H7 |
C2 |
H8 |
108.746 |
| H9 |
C3 |
H10 |
106.783 |
|
H11 |
C4 |
H12 |
106.532 |
| H13 |
C5 |
H14 |
107.891 |
|
H13 |
C5 |
H15 |
107.891 |
| H14 |
C5 |
H15 |
107.702 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
