Jump to
S1C2
Energy calculated at MP3/6-31+G**
| | hartrees |
|---|
| Energy at 0K | -217.781459 |
| Energy at 298.15K | -217.789341 |
| HF Energy | -217.131732 |
| Nuclear repulsion energy | 127.910371 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/6-31+G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3217 |
2994 |
28.05 |
|
|
|
| 2 |
A' |
3146 |
2927 |
61.25 |
|
|
|
| 3 |
A' |
3141 |
2923 |
0.72 |
|
|
|
| 4 |
A' |
3125 |
2908 |
21.13 |
|
|
|
| 5 |
A' |
1576 |
1467 |
1.32 |
|
|
|
| 6 |
A' |
1561 |
1452 |
5.60 |
|
|
|
| 7 |
A' |
1548 |
1440 |
0.85 |
|
|
|
| 8 |
A' |
1480 |
1378 |
15.43 |
|
|
|
| 9 |
A' |
1472 |
1370 |
6.13 |
|
|
|
| 10 |
A' |
1375 |
1279 |
2.21 |
|
|
|
| 11 |
A' |
1174 |
1092 |
1.55 |
|
|
|
| 12 |
A' |
1106 |
1029 |
78.28 |
|
|
|
| 13 |
A' |
1071 |
996 |
40.80 |
|
|
|
| 14 |
A' |
929 |
864 |
7.18 |
|
|
|
| 15 |
A' |
458 |
426 |
6.80 |
|
|
|
| 16 |
A' |
274 |
255 |
4.80 |
|
|
|
| 17 |
A" |
3215 |
2991 |
84.31 |
|
|
|
| 18 |
A" |
3200 |
2977 |
0.34 |
|
|
|
| 19 |
A" |
3182 |
2961 |
1.10 |
|
|
|
| 20 |
A" |
1553 |
1445 |
7.37 |
|
|
|
| 21 |
A" |
1351 |
1257 |
0.07 |
|
|
|
| 22 |
A" |
1304 |
1213 |
0.20 |
|
|
|
| 23 |
A" |
1228 |
1143 |
2.14 |
|
|
|
| 24 |
A" |
920 |
856 |
1.27 |
|
|
|
| 25 |
A" |
781 |
727 |
0.65 |
|
|
|
| 26 |
A" |
232 |
216 |
0.01 |
|
|
|
| 27 |
A" |
129 |
120 |
4.25 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21871.4 cm
-1
Scaled (by 0.9305) Zero Point Vibrational Energy (zpe) 20351.4 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3/6-31+G**
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.117 |
-0.781 |
0.000 |
| C2 |
0.000 |
0.729 |
0.000 |
| C3 |
-1.463 |
1.174 |
0.000 |
| F4 |
1.469 |
-1.155 |
0.000 |
| H5 |
-0.337 |
-1.220 |
0.889 |
| H6 |
-0.337 |
-1.220 |
-0.889 |
| H7 |
0.516 |
1.117 |
-0.878 |
| H8 |
0.516 |
1.117 |
0.878 |
| H9 |
-1.536 |
2.260 |
0.000 |
| H10 |
-1.986 |
0.804 |
-0.882 |
| H11 |
-1.986 |
0.804 |
0.882 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5140 | 2.5138 | 1.4030 | 1.0902 | 1.0902 | 2.1291 | 2.1291 | 3.4614 | 2.7776 | 2.7776 |
C2 | 1.5140 | | 1.5293 | 2.3891 | 2.1677 | 2.1677 | 1.0904 | 1.0904 | 2.1691 | 2.1746 | 2.1746 | C3 | 2.5138 | 1.5293 | | 3.7449 | 2.7910 | 2.7910 | 2.1660 | 2.1660 | 1.0883 | 1.0903 | 1.0903 | F4 | 1.4030 | 2.3891 | 3.7449 | | 2.0138 | 2.0138 | 2.6160 | 2.6160 | 4.5494 | 4.0692 | 4.0692 | H5 | 1.0902 | 2.1677 | 2.7910 | 2.0138 | | 1.7778 | 3.0513 | 2.4874 | 3.7865 | 3.1549 | 2.6111 | H6 | 1.0902 | 2.1677 | 2.7910 | 2.0138 | 1.7778 | | 2.4874 | 3.0513 | 3.7865 | 2.6111 | 3.1549 | H7 | 2.1291 | 1.0904 | 2.1660 | 2.6160 | 3.0513 | 2.4874 | | 1.7569 | 2.5079 | 2.5219 | 3.0756 | H8 | 2.1291 | 1.0904 | 2.1660 | 2.6160 | 2.4874 | 3.0513 | 1.7569 | | 2.5079 | 3.0756 | 2.5219 | H9 | 3.4614 | 2.1691 | 1.0883 | 4.5494 | 3.7865 | 3.7865 | 2.5079 | 2.5079 | | 1.7608 | 1.7608 | H10 | 2.7776 | 2.1746 | 1.0903 | 4.0692 | 3.1549 | 2.6111 | 2.5219 | 3.0756 | 1.7608 | | 1.7640 | H11 | 2.7776 | 2.1746 | 1.0903 | 4.0692 | 2.6111 | 3.1549 | 3.0756 | 2.5219 | 1.7608 | 1.7640 | |
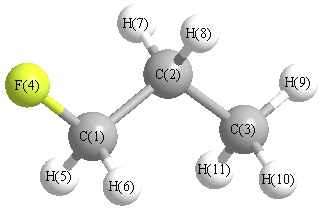 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
111.385 |
|
C1 |
C2 |
H7 |
108.577 |
| C1 |
C2 |
H8 |
108.577 |
|
C2 |
C1 |
F4 |
109.914 |
| C2 |
C1 |
H5 |
111.655 |
|
C2 |
C1 |
H6 |
111.655 |
| C2 |
C3 |
H9 |
110.800 |
|
C2 |
C3 |
H10 |
111.118 |
| C2 |
C3 |
H11 |
111.118 |
|
C3 |
C2 |
H7 |
110.424 |
| C3 |
C2 |
H8 |
110.424 |
|
F4 |
C1 |
H5 |
107.080 |
| F4 |
C1 |
H6 |
107.080 |
|
H5 |
C1 |
H6 |
109.250 |
| H7 |
C2 |
H8 |
107.335 |
|
H9 |
C3 |
H10 |
107.841 |
| H9 |
C3 |
H11 |
107.841 |
|
H10 |
C3 |
H11 |
107.978 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at MP3/6-31+G**
| | hartrees |
|---|
| Energy at 0K | -217.781896 |
| Energy at 298.15K | |
| HF Energy | -217.131843 |
| Nuclear repulsion energy | 130.270794 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/6-31+G**
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3226 |
3002 |
24.06 |
|
|
|
| 2 |
A |
3211 |
2988 |
59.50 |
|
|
|
| 3 |
A |
3206 |
2983 |
24.04 |
|
|
|
| 4 |
A |
3176 |
2955 |
16.50 |
|
|
|
| 5 |
A |
3146 |
2928 |
33.41 |
|
|
|
| 6 |
A |
3128 |
2911 |
19.80 |
|
|
|
| 7 |
A |
3125 |
2908 |
25.11 |
|
|
|
| 8 |
A |
1572 |
1463 |
0.65 |
|
|
|
| 9 |
A |
1559 |
1451 |
8.39 |
|
|
|
| 10 |
A |
1549 |
1441 |
6.81 |
|
|
|
| 11 |
A |
1531 |
1425 |
2.02 |
|
|
|
| 12 |
A |
1477 |
1374 |
7.24 |
|
|
|
| 13 |
A |
1466 |
1364 |
12.86 |
|
|
|
| 14 |
A |
1426 |
1327 |
1.89 |
|
|
|
| 15 |
A |
1336 |
1244 |
0.95 |
|
|
|
| 16 |
A |
1305 |
1215 |
1.02 |
|
|
|
| 17 |
A |
1213 |
1128 |
2.32 |
|
|
|
| 18 |
A |
1159 |
1078 |
11.14 |
|
|
|
| 19 |
A |
1128 |
1049 |
49.56 |
|
|
|
| 20 |
A |
1009 |
939 |
48.89 |
|
|
|
| 21 |
A |
946 |
881 |
3.12 |
|
|
|
| 22 |
A |
904 |
841 |
4.54 |
|
|
|
| 23 |
A |
790 |
735 |
0.85 |
|
|
|
| 24 |
A |
488 |
454 |
5.04 |
|
|
|
| 25 |
A |
322 |
300 |
1.27 |
|
|
|
| 26 |
A |
226 |
210 |
1.85 |
|
|
|
| 27 |
A |
145 |
135 |
3.04 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21883.5 cm
-1
Scaled (by 0.9305) Zero Point Vibrational Energy (zpe) 20362.6 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3/6-31+G**
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.793 |
0.520 |
0.301 |
| C2 |
-0.596 |
0.654 |
-0.287 |
| C3 |
-1.528 |
-0.485 |
0.120 |
| F4 |
1.397 |
-0.658 |
-0.171 |
| H5 |
1.441 |
1.346 |
0.010 |
| H6 |
0.765 |
0.444 |
1.388 |
| H7 |
-0.508 |
0.704 |
-1.374 |
| H8 |
-1.001 |
1.613 |
0.045 |
| H9 |
-2.516 |
-0.352 |
-0.317 |
| H10 |
-1.134 |
-1.443 |
-0.214 |
| H11 |
-1.641 |
-0.524 |
1.204 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5142 | 2.5355 | 1.4051 | 1.0890 | 1.0904 | 2.1286 | 2.1167 | 3.4772 | 2.7987 | 2.7984 |
C2 | 1.5142 | | 1.5274 | 2.3889 | 2.1709 | 2.1688 | 1.0910 | 1.0930 | 2.1680 | 2.1667 | 2.1693 | C3 | 2.5355 | 1.5274 | | 2.9444 | 3.4893 | 2.7803 | 2.1643 | 2.1646 | 1.0890 | 1.0881 | 1.0907 | F4 | 1.4051 | 2.3889 | 2.9444 | | 2.0121 | 2.0114 | 2.6325 | 3.3101 | 3.9280 | 2.6508 | 3.3378 | H5 | 1.0890 | 2.1709 | 3.4893 | 2.0121 | | 1.7799 | 2.4747 | 2.4568 | 4.3177 | 3.8023 | 3.7973 | H6 | 1.0904 | 2.1688 | 2.7803 | 2.0114 | 1.7799 | | 3.0524 | 2.5080 | 3.7826 | 3.1205 | 2.6005 | H7 | 2.1286 | 1.0910 | 2.1643 | 2.6325 | 2.4747 | 3.0524 | | 1.7562 | 2.5024 | 2.5192 | 3.0718 | H8 | 2.1167 | 1.0930 | 2.1646 | 3.3101 | 2.4568 | 2.5080 | 1.7562 | | 2.5070 | 3.0701 | 2.5138 | H9 | 3.4772 | 2.1680 | 1.0890 | 3.9280 | 4.3177 | 3.7826 | 2.5024 | 2.5070 | | 1.7642 | 1.7631 | H10 | 2.7987 | 2.1667 | 1.0881 | 2.6508 | 3.8023 | 3.1205 | 2.5192 | 3.0701 | 1.7642 | | 1.7642 | H11 | 2.7984 | 2.1693 | 1.0907 | 3.3378 | 3.7973 | 2.6005 | 3.0718 | 2.5138 | 1.7631 | 1.7642 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
112.940 |
|
C1 |
C2 |
H7 |
108.493 |
| C1 |
C2 |
H8 |
107.462 |
|
C2 |
C1 |
F4 |
109.776 |
| C2 |
C1 |
H5 |
111.977 |
|
C2 |
C1 |
H6 |
111.724 |
| C2 |
C3 |
H9 |
110.803 |
|
C2 |
C3 |
H10 |
110.749 |
| C2 |
C3 |
H11 |
110.803 |
|
C3 |
C2 |
H7 |
110.387 |
| C3 |
C2 |
H8 |
110.290 |
|
F4 |
C1 |
H5 |
106.869 |
| F4 |
C1 |
H6 |
106.737 |
|
H5 |
C1 |
H6 |
109.509 |
| H7 |
C2 |
H8 |
107.050 |
|
H9 |
C3 |
H10 |
108.256 |
| H9 |
C3 |
H11 |
107.981 |
|
H10 |
C3 |
H11 |
108.140 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
