All results from a given calculation for C4H9NO (Propanamide, 2-methyl-)
using model chemistry: MP3/6-31+G**
19 10 17 12 22
States and conformations
| State |
Conformation |
minimum conformation |
conformer description |
state description |
| 1 |
1 |
yes |
C1 |
1A |
Energy calculated at MP3/6-31+G**
| | hartrees |
|---|
| Energy at 0K | -287.018515 |
| Energy at 298.15K | |
| HF Energy | -286.064095 |
| Nuclear repulsion energy | 248.326321 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/6-31+G**
Geometric Data calculated at MP3/6-31+G**
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-1.349 |
-1.263 |
-0.026 |
| H2 |
-0.832 |
-2.161 |
-0.362 |
| H3 |
-1.406 |
-1.288 |
1.060 |
| H4 |
-2.359 |
-1.275 |
-0.433 |
| C5 |
-1.349 |
1.263 |
-0.025 |
| H6 |
-0.832 |
2.161 |
-0.362 |
| H7 |
-1.406 |
1.288 |
1.061 |
| C8 |
-0.610 |
0.000 |
-0.472 |
| H9 |
-0.535 |
0.000 |
-1.563 |
| N10 |
1.819 |
0.000 |
-0.755 |
| H11 |
2.757 |
0.000 |
-0.399 |
| H12 |
1.675 |
0.000 |
-1.745 |
| C13 |
0.788 |
0.000 |
0.139 |
| O14 |
0.960 |
-0.000 |
1.346 |
| H15 |
-2.359 |
1.275 |
-0.433 |
Atom - Atom Distances (Å)
| |
C1 |
H2 |
H3 |
H4 |
C5 |
H6 |
H7 |
C8 |
H9 |
N10 |
H11 |
H12 |
C13 |
O14 |
H15 |
| C1 | | 1.0895 | 1.0880 | 1.0894 | 2.5257 | 3.4790 | 2.7732 | 1.5297 | 2.1493 | 3.4875 | 4.3118 | 3.7010 | 2.4872 | 2.9678 | 2.7622 |
H2 | 1.0895 | | 1.7651 | 1.7666 | 3.4790 | 4.3222 | 3.7750 | 2.1753 | 2.4899 | 3.4429 | 4.1897 | 3.5877 | 2.7469 | 3.2865 | 3.7612 | H3 | 1.0880 | 1.7651 | | 1.7718 | 2.7732 | 3.7750 | 2.5764 | 2.1546 | 3.0494 | 3.9187 | 4.5954 | 4.3619 | 2.7055 | 2.7092 | 3.1163 | H4 | 1.0894 | 1.7666 | 1.7718 | | 2.7621 | 3.7612 | 3.1163 | 2.1652 | 2.4959 | 4.3804 | 5.2728 | 4.4301 | 3.4432 | 3.9761 | 2.5509 | C5 | 2.5257 | 3.4790 | 2.7732 | 2.7621 | | 1.0896 | 1.0880 | 1.5297 | 2.1493 | 3.4875 | 4.3117 | 3.7009 | 2.4872 | 2.9679 | 1.0894 | H6 | 3.4790 | 4.3222 | 3.7750 | 3.7612 | 1.0896 | | 1.7651 | 2.1753 | 2.4899 | 3.4430 | 4.1896 | 3.5874 | 2.7469 | 3.2866 | 1.7666 | H7 | 2.7732 | 3.7750 | 2.5764 | 3.1163 | 1.0880 | 1.7651 | | 2.1546 | 3.0494 | 3.9188 | 4.5953 | 4.3618 | 2.7055 | 2.7093 | 1.7718 | C8 | 1.5297 | 2.1753 | 2.1546 | 2.1652 | 1.5297 | 2.1753 | 2.1546 | | 1.0931 | 2.4454 | 3.3677 | 2.6159 | 1.5254 | 2.4023 | 2.1652 | H9 | 2.1493 | 2.4899 | 3.0494 | 2.4959 | 2.1493 | 2.4899 | 3.0494 | 1.0931 | | 2.4891 | 3.4921 | 2.2181 | 2.1556 | 3.2707 | 2.4959 | N10 | 3.4875 | 3.4429 | 3.9187 | 4.3804 | 3.4875 | 3.4430 | 3.9188 | 2.4454 | 2.4891 | | 1.0034 | 1.0006 | 1.3651 | 2.2697 | 4.3804 | H11 | 4.3118 | 4.1897 | 4.5954 | 5.2728 | 4.3117 | 4.1896 | 4.5953 | 3.3677 | 3.4921 | 1.0034 | | 1.7275 | 2.0414 | 2.5041 | 5.2727 | H12 | 3.7010 | 3.5877 | 4.3619 | 4.4301 | 3.7009 | 3.5874 | 4.3618 | 2.6159 | 2.2181 | 1.0006 | 1.7275 | | 2.0830 | 3.1728 | 4.4300 | C13 | 2.4872 | 2.7469 | 2.7055 | 3.4432 | 2.4872 | 2.7469 | 2.7055 | 1.5254 | 2.1556 | 1.3651 | 2.0414 | 2.0830 | | 1.2190 | 3.4432 | O14 | 2.9678 | 3.2865 | 2.7092 | 3.9761 | 2.9679 | 3.2866 | 2.7093 | 2.4023 | 3.2707 | 2.2697 | 2.5041 | 3.1728 | 1.2190 | | 3.9761 | H15 | 2.7622 | 3.7612 | 3.1163 | 2.5509 | 1.0894 | 1.7666 | 1.7718 | 2.1652 | 2.4959 | 4.3804 | 5.2727 | 4.4300 | 3.4432 | 3.9761 | |
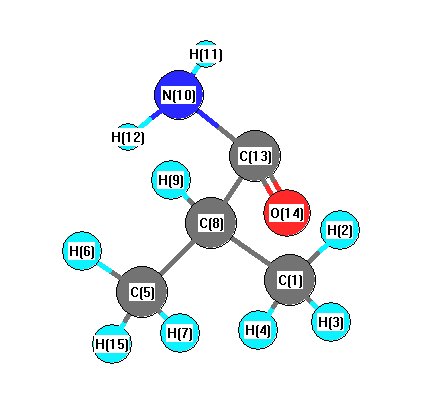 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C8 |
C5 |
111.282 |
|
C1 |
C8 |
H9 |
108.916 |
| C1 |
C8 |
C13 |
109.000 |
|
H2 |
C1 |
H3 |
108.308 |
| H2 |
C1 |
H4 |
108.341 |
|
H2 |
C1 |
C8 |
111.183 |
| H3 |
C1 |
H4 |
108.925 |
|
H3 |
C1 |
C8 |
109.633 |
| H4 |
C1 |
C8 |
110.390 |
|
C5 |
C8 |
H9 |
108.916 |
| C5 |
C8 |
C13 |
109.000 |
|
H6 |
C5 |
H7 |
108.308 |
| H6 |
C5 |
C8 |
111.184 |
|
H6 |
C5 |
H15 |
108.341 |
| H7 |
C5 |
C8 |
109.633 |
|
H7 |
C5 |
H15 |
108.925 |
| C8 |
C5 |
H15 |
110.390 |
|
C8 |
C13 |
N10 |
115.452 |
| C8 |
C13 |
O14 |
121.771 |
|
H9 |
C8 |
C13 |
109.712 |
| N10 |
C13 |
O14 |
122.777 |
|
H11 |
N10 |
H12 |
119.086 |
| H11 |
N10 |
C13 |
118.256 |
|
H12 |
N10 |
C13 |
122.658 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
