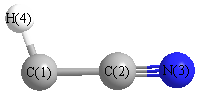Jump to
S1C2
S2C1
S2C2
Energy calculated at MP3/cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -131.036368 |
| Energy at 298.15K | -131.035898 |
| HF Energy | -130.690167 |
| Nuclear repulsion energy | 47.123032 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3364 |
3189 |
20.71 |
|
|
|
| 2 |
A' |
1977 |
1874 |
34.11 |
|
|
|
| 3 |
A' |
1125 |
1067 |
31.89 |
|
|
|
| 4 |
A' |
740 |
701 |
13.34 |
|
|
|
| 5 |
A' |
423 |
401 |
2.95 |
|
|
|
| 6 |
A" |
486 |
460 |
3.43 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 4057.1 cm
-1
Scaled (by 0.9479) Zero Point Vibrational Energy (zpe) 3845.7 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3/cc-pVDZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-0.114 |
-1.249 |
0.000 |
| C2 |
0.000 |
0.110 |
0.000 |
| N3 |
0.021 |
1.280 |
0.000 |
| H4 |
0.533 |
-2.123 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
N3 |
H4 |
| C1 | | 1.3645 | 2.5326 | 1.0867 |
C2 | 1.3645 | | 1.1694 | 2.2959 | N3 | 2.5326 | 1.1694 | | 3.4406 | H4 | 1.0867 | 2.2959 | 3.4406 | |
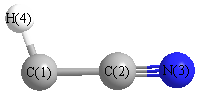 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
N3 |
176.265 |
|
C2 |
C1 |
H4 |
138.700 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
S2C1
S2C2
Energy calculated at MP3/cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -131.033008 |
| Energy at 298.15K | |
| HF Energy | -130.688960 |
| Nuclear repulsion energy | 47.290047 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
Σ |
3484 |
3303 |
78.69 |
|
|
|
| 2 |
Σ |
1572 |
1490 |
63.11 |
|
|
|
| 3 |
Σ |
1256 |
1191 |
40.02 |
|
|
|
| 4 |
Π |
479 |
454 |
0.13 |
|
|
|
| 4 |
Π |
479 |
454 |
0.13 |
|
|
|
| 5 |
Π |
421i |
399i |
47.73 |
|
|
|
| 5 |
Π |
421i |
399i |
47.73 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 3214.1 cm
-1
Scaled (by 0.9479) Zero Point Vibrational Energy (zpe) 3046.6 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3/cc-pVDZ
Point Group is C∞v
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.000 |
0.000 |
-1.216 |
| C2 |
0.000 |
0.000 |
0.093 |
| N3 |
0.000 |
0.000 |
1.291 |
| H4 |
0.000 |
0.000 |
-2.292 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
N3 |
H4 |
| C1 | | 1.3093 | 2.5069 | 1.0758 |
C2 | 1.3093 | | 1.1976 | 2.3851 | N3 | 2.5069 | 1.1976 | | 3.5828 | H4 | 1.0758 | 2.3851 | 3.5828 | |
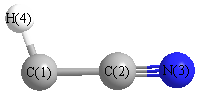 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
N3 |
180.000 |
|
C2 |
C1 |
H4 |
180.000 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Charges from optimized geometry at MP3/cc-pVDZ
Charges (e)
| Number |
Element |
Mulliken |
CHELPG |
AIM |
ESP |
| 1 |
C |
-0.018 |
|
|
|
| 2 |
C |
0.021 |
|
|
|
| 3 |
N |
-0.111 |
|
|
|
| 4 |
H |
0.107 |
|
|
|
Electric dipole moments
Electric Quadrupole moment
Quadrupole components in D Å
Polarizabilities
Components of the polarizability tensor.
Units are
Å
3 (Angstrom cubed)
Change units.
| |
x |
y |
z |
| x |
0.000 |
0.000 |
0.000 |
| y |
0.000 |
0.000 |
0.000 |
| z |
0.000 |
0.000 |
0.000 |
<r2> (average value of r
2) Å
2
| <r2> |
36.292 |
| (<r2>)1/2 |
6.024 |
Jump to
S1C1
S1C2
S2C2
Energy calculated at MP3/cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -131.022576 |
| Energy at 298.15K | -131.022008 |
| HF Energy | -130.629915 |
| Nuclear repulsion energy | 46.465147 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/cc-pVDZ
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3081 |
2920 |
18.20 |
|
|
|
| 2 |
A' |
2248 |
2131 |
30.48 |
|
|
|
| 3 |
A' |
1073 |
1017 |
66.46 |
|
|
|
| 4 |
A' |
998 |
946 |
7.69 |
|
|
|
| 5 |
A' |
422 |
400 |
17.24 |
|
|
|
| 6 |
A" |
317 |
301 |
8.99 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 4069.2 cm
-1
Scaled (by 0.9479) Zero Point Vibrational Energy (zpe) 3857.2 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3/cc-pVDZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.004 |
-1.314 |
0.000 |
| C2 |
0.000 |
0.104 |
0.000 |
| N3 |
-0.156 |
1.271 |
0.000 |
| H4 |
1.072 |
-1.635 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
N3 |
H4 |
| C1 | | 1.4184 | 2.5902 | 1.1151 |
C2 | 1.4184 | | 1.1773 | 2.0427 | N3 | 2.5902 | 1.1773 | | 3.1547 | H4 | 1.1151 | 2.0427 | 3.1547 | |
 More geometry information
More geometry information
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
S1C2
S2C1
Energy calculated at MP3/cc-pVDZ
| | hartrees |
|---|
| Energy at 0K | -131.022576 |
| Energy at 298.15K | -131.022008 |
| HF Energy | -130.629915 |
| Nuclear repulsion energy | 46.465147 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3/cc-pVDZ
Geometric Data calculated at MP3/cc-pVDZ
Point Group is Cs
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability