All results from a given calculation for C4H10O (Methyl propyl ether)
using model chemistry: CCSD(T)=FULL/cc-pVTZ
19 10 17 12 22
States and conformations
| State |
Conformation |
minimum conformation |
conformer description |
state description |
| 1 |
1 |
yes |
C2 |
1A' |
Energy calculated at CCSD(T)=FULL/cc-pVTZ
| | hartrees |
|---|
| Energy at 0K | -232.898872 |
| Energy at 298.15K | |
| HF Energy | -232.227256 |
| Nuclear repulsion energy | 189.591468 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at CCSD(T)=FULL/cc-pVTZ
Geometric Data calculated at CCSD(T)=FULL/cc-pVTZ
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
-2.298 |
0.669 |
0.000 |
| O2 |
-1.270 |
-0.291 |
0.000 |
| C3 |
0.000 |
0.320 |
0.000 |
| C4 |
1.063 |
-0.753 |
0.000 |
| C5 |
2.464 |
-0.161 |
0.000 |
| H6 |
-3.245 |
0.138 |
0.000 |
| H7 |
-2.248 |
1.307 |
0.887 |
| H8 |
-2.248 |
1.307 |
-0.887 |
| H9 |
0.111 |
0.960 |
0.883 |
| H10 |
0.111 |
0.960 |
-0.883 |
| H11 |
0.917 |
-1.384 |
-0.875 |
| H12 |
0.917 |
-1.384 |
0.875 |
| H13 |
2.626 |
0.461 |
0.880 |
| H14 |
2.626 |
0.461 |
-0.880 |
| H15 |
3.220 |
-0.942 |
0.000 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
C3 |
C4 |
C5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
H12 |
H13 |
H14 |
H15 |
| C1 | | 1.4071 | 2.3248 | 3.6498 | 4.8343 | 1.0857 | 1.0942 | 1.0942 | 2.5829 | 2.5829 | 3.9144 | 3.9144 | 5.0070 | 5.0070 | 5.7492 |
O2 | 1.4071 | | 1.4094 | 2.3780 | 3.7363 | 2.0216 | 2.0735 | 2.0735 | 2.0626 | 2.0626 | 2.5970 | 2.5970 | 4.0644 | 4.0644 | 4.5372 | C3 | 2.3248 | 1.4094 | | 1.5107 | 2.5106 | 3.2505 | 2.6107 | 2.6107 | 1.0967 | 1.0967 | 2.1243 | 2.1243 | 2.7733 | 2.7733 | 3.4589 | C4 | 3.6498 | 2.3780 | 1.5107 | | 1.5215 | 4.3995 | 3.9995 | 3.9995 | 2.1503 | 2.1503 | 1.0889 | 1.0889 | 2.1663 | 2.1663 | 2.1658 | C5 | 4.8343 | 3.7363 | 2.5106 | 1.5215 | | 5.7174 | 5.0147 | 5.0147 | 2.7522 | 2.7522 | 2.1579 | 2.1579 | 1.0895 | 1.0895 | 1.0874 | H6 | 1.0857 | 2.0216 | 3.2505 | 4.3995 | 5.7174 | | 1.7746 | 1.7746 | 3.5669 | 3.5669 | 4.5178 | 4.5178 | 5.9460 | 5.9460 | 6.5554 | H7 | 1.0942 | 2.0735 | 2.6107 | 3.9995 | 5.0147 | 1.7746 | | 1.7745 | 2.3845 | 2.9700 | 4.5133 | 4.1549 | 4.9473 | 5.2535 | 5.9792 | H8 | 1.0942 | 2.0735 | 2.6107 | 3.9995 | 5.0147 | 1.7746 | 1.7745 | | 2.9700 | 2.3845 | 4.1549 | 4.5133 | 5.2535 | 4.9473 | 5.9792 | H9 | 2.5829 | 2.0626 | 1.0967 | 2.1503 | 2.7522 | 3.5669 | 2.3845 | 2.9700 | | 1.7668 | 3.0400 | 2.4795 | 2.5643 | 3.1122 | 3.7508 | H10 | 2.5829 | 2.0626 | 1.0967 | 2.1503 | 2.7522 | 3.5669 | 2.9700 | 2.3845 | 1.7668 | | 2.4795 | 3.0400 | 3.1122 | 2.5643 | 3.7508 | H11 | 3.9144 | 2.5970 | 2.1243 | 1.0889 | 2.1579 | 4.5178 | 4.5133 | 4.1549 | 3.0400 | 2.4795 | | 1.7509 | 3.0671 | 2.5148 | 2.5034 | H12 | 3.9144 | 2.5970 | 2.1243 | 1.0889 | 2.1579 | 4.5178 | 4.1549 | 4.5133 | 2.4795 | 3.0400 | 1.7509 | | 2.5148 | 3.0671 | 2.5034 | H13 | 5.0070 | 4.0644 | 2.7733 | 2.1663 | 1.0895 | 5.9460 | 4.9473 | 5.2535 | 2.5643 | 3.1122 | 3.0671 | 2.5148 | | 1.7605 | 1.7594 | H14 | 5.0070 | 4.0644 | 2.7733 | 2.1663 | 1.0895 | 5.9460 | 5.2535 | 4.9473 | 3.1122 | 2.5643 | 2.5148 | 3.0671 | 1.7605 | | 1.7594 | H15 | 5.7492 | 4.5372 | 3.4589 | 2.1658 | 1.0874 | 6.5554 | 5.9792 | 5.9792 | 3.7508 | 3.7508 | 2.5034 | 2.5034 | 1.7594 | 1.7594 | |
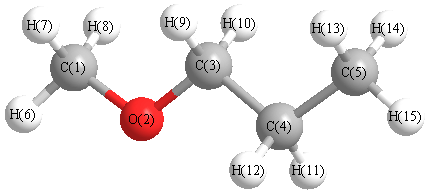 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
O2 |
C3 |
111.262 |
|
O2 |
C1 |
H6 |
107.689 |
| O2 |
C1 |
H7 |
111.373 |
|
O2 |
C1 |
H8 |
111.373 |
| O2 |
C3 |
C4 |
109.003 |
|
O2 |
C3 |
H9 |
110.154 |
| O2 |
C3 |
H10 |
110.154 |
|
C3 |
C4 |
C5 |
111.785 |
| C3 |
C4 |
H11 |
108.518 |
|
C3 |
C4 |
H12 |
108.518 |
| C4 |
C3 |
H9 |
110.104 |
|
C4 |
C3 |
H10 |
110.104 |
| C4 |
C5 |
H13 |
111.046 |
|
C4 |
C5 |
H14 |
111.046 |
| C4 |
C5 |
H15 |
111.137 |
|
C5 |
C4 |
H11 |
110.420 |
| C5 |
C4 |
H12 |
110.420 |
|
H6 |
C1 |
H7 |
108.995 |
| H6 |
C1 |
H8 |
108.995 |
|
H7 |
C1 |
H8 |
108.366 |
| H9 |
C3 |
H10 |
107.311 |
|
H11 |
C4 |
H12 |
107.032 |
| H13 |
C5 |
H14 |
107.779 |
|
H13 |
C5 |
H15 |
107.840 |
| H14 |
C5 |
H15 |
107.840 |
|
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
