Jump to
S1C2
S1C3
Energy calculated at MP3=FULL/6-31G(2df,p)
| | hartrees |
|---|
| Energy at 0K | -208.767496 |
| Energy at 298.15K | -208.773593 |
| HF Energy | -207.984384 |
| Nuclear repulsion energy | 120.037530 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3=FULL/6-31G(2df,p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3732 |
3732 |
40.34 |
|
|
|
| 2 |
A' |
3217 |
3217 |
11.21 |
|
|
|
| 3 |
A' |
3117 |
3117 |
40.38 |
|
|
|
| 4 |
A' |
3034 |
3034 |
63.79 |
|
|
|
| 5 |
A' |
1901 |
1901 |
499.09 |
|
|
|
| 6 |
A' |
1585 |
1585 |
20.38 |
|
|
|
| 7 |
A' |
1537 |
1537 |
16.22 |
|
|
|
| 8 |
A' |
1516 |
1516 |
10.49 |
|
|
|
| 9 |
A' |
1449 |
1449 |
14.10 |
|
|
|
| 10 |
A' |
1346 |
1346 |
118.23 |
|
|
|
| 11 |
A' |
1212 |
1212 |
23.48 |
|
|
|
| 12 |
A' |
1044 |
1044 |
47.68 |
|
|
|
| 13 |
A' |
632 |
632 |
14.88 |
|
|
|
| 14 |
A' |
353 |
353 |
8.16 |
|
|
|
| 15 |
A" |
3183 |
3183 |
21.12 |
|
|
|
| 16 |
A" |
1538 |
1538 |
5.40 |
|
|
|
| 17 |
A" |
1191 |
1191 |
0.49 |
|
|
|
| 18 |
A" |
1102 |
1102 |
0.02 |
|
|
|
| 19 |
A" |
620 |
620 |
120.30 |
|
|
|
| 20 |
A" |
206 |
206 |
0.90 |
|
|
|
| 21 |
A" |
93 |
93 |
0.92 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 16802.9 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 16802.9 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3=FULL/6-31G(2df,p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.290 |
-0.749 |
0.000 |
| O2 |
1.397 |
-1.217 |
0.000 |
| N3 |
0.000 |
0.570 |
0.000 |
| C4 |
-1.333 |
1.114 |
0.000 |
| H5 |
-0.621 |
-1.370 |
0.000 |
| H6 |
0.787 |
1.193 |
0.000 |
| H7 |
-2.048 |
0.294 |
0.000 |
| H8 |
-1.520 |
1.721 |
0.885 |
| H9 |
-1.520 |
1.721 |
-0.885 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
N3 |
C4 |
H5 |
H6 |
H7 |
H8 |
H9 |
| C1 | | 1.2016 | 1.3506 | 2.4708 | 1.1024 | 2.0042 | 2.5601 | 3.1878 | 3.1878 |
O2 | 1.2016 | | 2.2682 | 3.5896 | 2.0235 | 2.4854 | 3.7616 | 4.2337 | 4.2337 | N3 | 1.3506 | 2.2682 | | 1.4397 | 2.0368 | 1.0035 | 2.0663 | 2.1020 | 2.1020 | C4 | 2.4708 | 3.5896 | 1.4397 | | 2.5839 | 2.1216 | 1.0876 | 1.0896 | 1.0896 | H5 | 1.1024 | 2.0235 | 2.0368 | 2.5839 | | 2.9238 | 2.1921 | 3.3388 | 3.3388 | H6 | 2.0042 | 2.4854 | 1.0035 | 2.1216 | 2.9238 | | 2.9739 | 2.5267 | 2.5267 | H7 | 2.5601 | 3.7616 | 2.0663 | 1.0876 | 2.1921 | 2.9739 | | 1.7605 | 1.7605 | H8 | 3.1878 | 4.2337 | 2.1020 | 1.0896 | 3.3388 | 2.5267 | 1.7605 | | 1.7699 | H9 | 3.1878 | 4.2337 | 2.1020 | 1.0896 | 3.3388 | 2.5267 | 1.7605 | 1.7699 | |
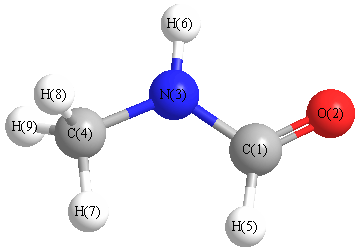 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
N3 |
C4 |
124.600 |
|
C1 |
N3 |
H6 |
115.935 |
| O2 |
C1 |
N3 |
125.322 |
|
O2 |
C1 |
H5 |
122.806 |
| N3 |
C1 |
H5 |
111.873 |
|
N3 |
C4 |
H7 |
108.894 |
| N3 |
C4 |
H8 |
111.672 |
|
N3 |
C4 |
H9 |
111.672 |
| C4 |
N3 |
H6 |
119.465 |
|
H7 |
C4 |
H8 |
107.922 |
| H7 |
C4 |
H9 |
107.922 |
|
H8 |
C4 |
H9 |
108.620 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
S1C3
Energy calculated at MP3=FULL/6-31G(2df,p)
| | hartrees |
|---|
| Energy at 0K | -208.767496 |
| Energy at 298.15K | -208.773593 |
| HF Energy | -207.984384 |
| Nuclear repulsion energy | 120.037530 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3=FULL/6-31G(2df,p)
Geometric Data calculated at MP3=FULL/6-31G(2df,p)
Point Group is Cs
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
S1C2
Energy calculated at MP3=FULL/6-31G(2df,p)
| | hartrees |
|---|
| Energy at 0K | -208.769274 |
| Energy at 298.15K | -208.775123 |
| HF Energy | -207.985884 |
| Nuclear repulsion energy | 122.493853 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3=FULL/6-31G(2df,p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3781 |
3781 |
49.45 |
|
|
|
| 2 |
A |
3235 |
3235 |
7.76 |
|
|
|
| 3 |
A |
3204 |
3204 |
14.53 |
|
|
|
| 4 |
A |
3128 |
3128 |
24.94 |
|
|
|
| 5 |
A |
3051 |
3051 |
84.86 |
|
|
|
| 6 |
A |
1883 |
1883 |
332.12 |
|
|
|
| 7 |
A |
1592 |
1592 |
121.18 |
|
|
|
| 8 |
A |
1554 |
1554 |
27.93 |
|
|
|
| 9 |
A |
1533 |
1533 |
18.01 |
|
|
|
| 10 |
A |
1498 |
1498 |
12.65 |
|
|
|
| 11 |
A |
1469 |
1469 |
3.66 |
|
|
|
| 12 |
A |
1283 |
1283 |
55.20 |
|
|
|
| 13 |
A |
1202 |
1202 |
2.39 |
|
|
|
| 14 |
A |
1193 |
1193 |
2.52 |
|
|
|
| 15 |
A |
1087 |
1087 |
0.76 |
|
|
|
| 16 |
A |
1007 |
1007 |
17.07 |
|
|
|
| 17 |
A |
795 |
795 |
7.63 |
|
|
|
| 18 |
A |
486 |
486 |
19.13 |
|
|
|
| 19 |
A |
279 |
279 |
12.05 |
|
|
|
| 20 |
A |
224 |
224 |
89.77 |
|
|
|
| 21 |
A |
13 |
13 |
4.49 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 16747.4 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 16747.4 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3=FULL/6-31G(2df,p)
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.860 |
0.428 |
0.004 |
| O2 |
1.364 |
-0.665 |
0.000 |
| N3 |
-0.469 |
0.657 |
-0.020 |
| C4 |
-1.411 |
-0.439 |
0.005 |
| H5 |
1.446 |
1.359 |
0.016 |
| H6 |
-0.799 |
1.597 |
0.059 |
| H7 |
-2.404 |
-0.060 |
-0.220 |
| H8 |
-1.135 |
-1.177 |
-0.744 |
| H9 |
-1.430 |
-0.933 |
0.975 |
Atom - Atom Distances (Å)
| |
C1 |
O2 |
N3 |
C4 |
H5 |
H6 |
H7 |
H8 |
H9 |
| C1 | | 1.2039 | 1.3485 | 2.4303 | 1.1007 | 2.0305 | 3.3076 | 2.6665 | 2.8352 |
O2 | 1.2039 | | 2.2607 | 2.7847 | 2.0261 | 3.1312 | 3.8232 | 2.6572 | 2.9719 | N3 | 1.3485 | 2.2607 | | 1.4454 | 2.0397 | 0.9994 | 2.0736 | 2.0812 | 2.1078 | C4 | 2.4303 | 2.7847 | 1.4454 | | 3.3755 | 2.1266 | 1.0863 | 1.0869 | 1.0896 | H5 | 1.1007 | 2.0261 | 2.0397 | 3.3755 | | 2.2581 | 4.1101 | 3.6970 | 3.8006 | H6 | 2.0305 | 3.1312 | 0.9994 | 2.1266 | 2.2581 | | 2.3239 | 2.9072 | 2.7642 | H7 | 3.3076 | 3.8232 | 2.0736 | 1.0863 | 4.1101 | 2.3239 | | 1.7700 | 1.7719 | H8 | 2.6665 | 2.6572 | 2.0812 | 1.0869 | 3.6970 | 2.9072 | 1.7700 | | 1.7614 | H9 | 2.8352 | 2.9719 | 2.1078 | 1.0896 | 3.8006 | 2.7642 | 1.7719 | 1.7614 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
N3 |
C4 |
120.847 |
|
C1 |
N3 |
H6 |
118.978 |
| O2 |
C1 |
N3 |
124.590 |
|
O2 |
C1 |
H5 |
123.024 |
| N3 |
C1 |
H5 |
112.383 |
|
N3 |
C4 |
H7 |
109.161 |
| N3 |
C4 |
H8 |
109.735 |
|
N3 |
C4 |
H9 |
111.731 |
| C4 |
N3 |
H6 |
119.763 |
|
H7 |
C4 |
H8 |
109.076 |
| H7 |
C4 |
H9 |
109.041 |
|
H8 |
C4 |
H9 |
108.053 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
