Jump to
S1C2
Energy calculated at MP3=FULL/6-31G(2df,p)
| | hartrees |
|---|
| Energy at 0K | -217.920960 |
| Energy at 298.15K | -217.928893 |
| HF Energy | -217.132885 |
| Nuclear repulsion energy | 128.829014 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3=FULL/6-31G(2df,p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A' |
3212 |
3212 |
18.09 |
|
|
|
| 2 |
A' |
3136 |
3136 |
23.67 |
|
|
|
| 3 |
A' |
3123 |
3123 |
15.63 |
|
|
|
| 4 |
A' |
3109 |
3109 |
24.74 |
|
|
|
| 5 |
A' |
1572 |
1572 |
3.09 |
|
|
|
| 6 |
A' |
1558 |
1558 |
4.24 |
|
|
|
| 7 |
A' |
1543 |
1543 |
0.17 |
|
|
|
| 8 |
A' |
1488 |
1488 |
17.05 |
|
|
|
| 9 |
A' |
1458 |
1458 |
2.51 |
|
|
|
| 10 |
A' |
1372 |
1372 |
1.08 |
|
|
|
| 11 |
A' |
1176 |
1176 |
10.52 |
|
|
|
| 12 |
A' |
1150 |
1150 |
82.44 |
|
|
|
| 13 |
A' |
1084 |
1084 |
3.33 |
|
|
|
| 14 |
A' |
930 |
930 |
7.57 |
|
|
|
| 15 |
A' |
463 |
463 |
5.53 |
|
|
|
| 16 |
A' |
272 |
272 |
2.80 |
|
|
|
| 17 |
A" |
3206 |
3206 |
44.40 |
|
|
|
| 18 |
A" |
3184 |
3184 |
7.80 |
|
|
|
| 19 |
A" |
3155 |
3155 |
17.90 |
|
|
|
| 20 |
A" |
1554 |
1554 |
6.64 |
|
|
|
| 21 |
A" |
1367 |
1367 |
0.01 |
|
|
|
| 22 |
A" |
1319 |
1319 |
0.92 |
|
|
|
| 23 |
A" |
1234 |
1234 |
1.22 |
|
|
|
| 24 |
A" |
920 |
920 |
1.80 |
|
|
|
| 25 |
A" |
781 |
781 |
1.63 |
|
|
|
| 26 |
A" |
241 |
241 |
0.00 |
|
|
|
| 27 |
A" |
142 |
142 |
2.77 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21873.8 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 21873.8 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3=FULL/6-31G(2df,p)
Point Group is Cs
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.137 |
-0.778 |
0.000 |
| C2 |
0.000 |
0.728 |
0.000 |
| C3 |
-1.465 |
1.149 |
0.000 |
| F4 |
1.460 |
-1.132 |
0.000 |
| H5 |
-0.336 |
-1.210 |
0.884 |
| H6 |
-0.336 |
-1.210 |
-0.884 |
| H7 |
0.512 |
1.124 |
-0.877 |
| H8 |
0.512 |
1.124 |
0.877 |
| H9 |
-1.560 |
2.232 |
0.000 |
| H10 |
-1.983 |
0.770 |
-0.880 |
| H11 |
-1.983 |
0.770 |
0.880 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5121 | 2.5058 | 1.3701 | 1.0918 | 1.0918 | 2.1275 | 2.1275 | 3.4557 | 2.7691 | 2.7691 |
C2 | 1.5121 | | 1.5243 | 2.3649 | 2.1562 | 2.1562 | 1.0898 | 1.0898 | 2.1673 | 2.1704 | 2.1704 | C3 | 2.5058 | 1.5243 | | 3.7097 | 2.7602 | 2.7602 | 2.1629 | 2.1629 | 1.0876 | 1.0895 | 1.0895 | F4 | 1.3701 | 2.3649 | 3.7097 | | 2.0041 | 2.0041 | 2.5995 | 2.5995 | 4.5215 | 4.0317 | 4.0317 | H5 | 1.0918 | 2.1562 | 2.7602 | 2.0041 | | 1.7683 | 3.0439 | 2.4829 | 3.7586 | 3.1219 | 2.5755 | H6 | 1.0918 | 2.1562 | 2.7602 | 2.0041 | 1.7683 | | 2.4829 | 3.0439 | 3.7586 | 2.5755 | 3.1219 | H7 | 2.1275 | 1.0898 | 2.1629 | 2.5995 | 3.0439 | 2.4829 | | 1.7536 | 2.5083 | 2.5203 | 3.0724 | H8 | 2.1275 | 1.0898 | 2.1629 | 2.5995 | 2.4829 | 3.0439 | 1.7536 | | 2.5083 | 3.0724 | 2.5203 | H9 | 3.4557 | 2.1673 | 1.0876 | 4.5215 | 3.7586 | 3.7586 | 2.5083 | 2.5083 | | 1.7584 | 1.7584 | H10 | 2.7691 | 2.1704 | 1.0895 | 4.0317 | 3.1219 | 2.5755 | 2.5203 | 3.0724 | 1.7584 | | 1.7607 | H11 | 2.7691 | 2.1704 | 1.0895 | 4.0317 | 2.5755 | 3.1219 | 3.0724 | 2.5203 | 1.7584 | 1.7607 | |
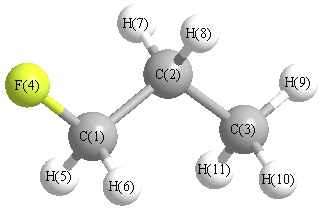 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
111.224 |
|
C1 |
C2 |
H7 |
108.614 |
| C1 |
C2 |
H8 |
108.614 |
|
C2 |
C1 |
F4 |
110.173 |
| C2 |
C1 |
H5 |
110.765 |
|
C2 |
C1 |
H6 |
110.765 |
| C2 |
C3 |
H9 |
111.045 |
|
C2 |
C3 |
H10 |
111.180 |
| C2 |
C3 |
H11 |
111.180 |
|
C3 |
C2 |
H7 |
110.564 |
| C3 |
C2 |
H8 |
110.564 |
|
F4 |
C1 |
H5 |
108.452 |
| F4 |
C1 |
H6 |
108.452 |
|
H5 |
C1 |
H6 |
108.153 |
| H7 |
C2 |
H8 |
107.137 |
|
H9 |
C3 |
H10 |
107.733 |
| H9 |
C3 |
H11 |
107.733 |
|
H10 |
C3 |
H11 |
107.805 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
Jump to
S1C1
Energy calculated at MP3=FULL/6-31G(2df,p)
| | hartrees |
|---|
| Energy at 0K | -217.921734 |
| Energy at 298.15K | |
| HF Energy | -217.133200 |
| Nuclear repulsion energy | 131.440191 |
The energy at 298.15K was derived from the energy at 0K
and an integrated heat capacity that used the calculated vibrational frequencies.
Vibrational Frequencies calculated at MP3=FULL/6-31G(2df,p)
| Mode Number |
Symmetry |
Frequency
(cm-1) |
Scaled Frequency
(cm-1) |
IR Intensities
(km mol-1) |
Raman Act
(Å4/u) |
Dep P |
Dep U |
|---|
| 1 |
A |
3224 |
3224 |
15.02 |
|
|
|
| 2 |
A |
3207 |
3207 |
28.31 |
|
|
|
| 3 |
A |
3177 |
3177 |
24.19 |
|
|
|
| 4 |
A |
3164 |
3164 |
28.82 |
|
|
|
| 5 |
A |
3127 |
3127 |
15.14 |
|
|
|
| 6 |
A |
3126 |
3126 |
12.96 |
|
|
|
| 7 |
A |
3113 |
3113 |
31.70 |
|
|
|
| 8 |
A |
1567 |
1567 |
1.22 |
|
|
|
| 9 |
A |
1559 |
1559 |
6.99 |
|
|
|
| 10 |
A |
1547 |
1547 |
5.67 |
|
|
|
| 11 |
A |
1526 |
1526 |
2.08 |
|
|
|
| 12 |
A |
1479 |
1479 |
14.15 |
|
|
|
| 13 |
A |
1455 |
1455 |
4.21 |
|
|
|
| 14 |
A |
1419 |
1419 |
0.34 |
|
|
|
| 15 |
A |
1346 |
1346 |
1.45 |
|
|
|
| 16 |
A |
1324 |
1324 |
1.72 |
|
|
|
| 17 |
A |
1216 |
1216 |
1.34 |
|
|
|
| 18 |
A |
1177 |
1177 |
42.38 |
|
|
|
| 19 |
A |
1142 |
1142 |
21.67 |
|
|
|
| 20 |
A |
1024 |
1024 |
26.07 |
|
|
|
| 21 |
A |
949 |
949 |
2.57 |
|
|
|
| 22 |
A |
911 |
911 |
3.19 |
|
|
|
| 23 |
A |
792 |
792 |
0.55 |
|
|
|
| 24 |
A |
491 |
491 |
3.15 |
|
|
|
| 25 |
A |
323 |
323 |
1.14 |
|
|
|
| 26 |
A |
233 |
233 |
1.81 |
|
|
|
| 27 |
A |
150 |
150 |
2.10 |
|
|
|
Unscaled Zero Point Vibrational Energy (zpe) 21883.5 cm
-1
Scaled (by 1) Zero Point Vibrational Energy (zpe) 21883.5 cm
-1
See section
III.C.1 List or set vibrational scaling factors
to change the scale factors used here.
See section
III.C.2
Calculate a vibrational scaling factor for a given set of molecules
to determine the least squares best scaling factor.
Geometric Data calculated at MP3=FULL/6-31G(2df,p)
Point Group is C1
Cartesians (Å)
| Atom |
x (Å) |
y (Å) |
z (Å) |
|---|
| C1 |
0.802 |
0.505 |
0.293 |
| C2 |
-0.589 |
0.669 |
-0.278 |
| C3 |
-1.503 |
-0.484 |
0.118 |
| F4 |
1.358 |
-0.660 |
-0.173 |
| H5 |
1.451 |
1.332 |
0.005 |
| H6 |
0.770 |
0.452 |
1.384 |
| H7 |
-0.512 |
0.734 |
-1.364 |
| H8 |
-0.992 |
1.620 |
0.073 |
| H9 |
-2.491 |
-0.375 |
-0.324 |
| H10 |
-1.083 |
-1.430 |
-0.215 |
| H11 |
-1.622 |
-0.531 |
1.200 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
C3 |
F4 |
H5 |
H6 |
H7 |
H8 |
H9 |
H10 |
H11 |
| C1 | | 1.5123 | 2.5143 | 1.3729 | 1.0907 | 1.0919 | 2.1267 | 2.1239 | 3.4640 | 2.7485 | 2.7880 |
C2 | 1.5123 | | 1.5241 | 2.3597 | 2.1635 | 2.1571 | 1.0902 | 1.0910 | 2.1706 | 2.1574 | 2.1668 | C3 | 2.5143 | 1.5241 | | 2.8814 | 3.4699 | 2.7647 | 2.1591 | 2.1655 | 1.0879 | 1.0874 | 1.0897 | F4 | 1.3729 | 2.3597 | 2.8814 | | 2.0026 | 2.0018 | 2.6184 | 3.2840 | 3.8628 | 2.5597 | 3.2842 | H5 | 1.0907 | 2.1635 | 3.4699 | 2.0026 | | 1.7722 | 2.4662 | 2.4615 | 4.3086 | 3.7549 | 3.7881 | H6 | 1.0919 | 2.1571 | 2.7647 | 2.0018 | 1.7722 | | 3.0442 | 2.4871 | 3.7726 | 3.0870 | 2.5929 | H7 | 2.1267 | 1.0902 | 2.1591 | 2.6184 | 2.4662 | 3.0442 | | 1.7551 | 2.4958 | 2.5156 | 3.0672 | H8 | 2.1239 | 1.0910 | 2.1655 | 3.2840 | 2.4615 | 2.4871 | 1.7551 | | 2.5264 | 3.0648 | 2.5091 | H9 | 3.4640 | 2.1706 | 1.0879 | 3.8628 | 4.3086 | 3.7726 | 2.4958 | 2.5264 | | 1.7632 | 1.7614 | H10 | 2.7485 | 2.1574 | 1.0874 | 2.5597 | 3.7549 | 3.0870 | 2.5156 | 3.0648 | 1.7632 | | 1.7610 | H11 | 2.7880 | 2.1668 | 1.0897 | 3.2842 | 3.7881 | 2.5929 | 3.0672 | 2.5091 | 1.7614 | 1.7610 | |
 More geometry information
More geometry information
Calculated Bond Angles
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| C1 |
C2 |
C3 |
111.795 |
|
C1 |
C2 |
H7 |
108.521 |
| C1 |
C2 |
H8 |
108.258 |
|
C2 |
C1 |
F4 |
109.649 |
| C2 |
C1 |
H5 |
111.406 |
|
C2 |
C1 |
H6 |
110.821 |
| C2 |
C3 |
H9 |
111.304 |
|
C2 |
C3 |
H10 |
110.287 |
| C2 |
C3 |
H11 |
110.895 |
|
C3 |
C2 |
H7 |
110.252 |
| C3 |
C2 |
H8 |
110.712 |
|
F4 |
C1 |
H5 |
108.211 |
| F4 |
C1 |
H6 |
108.077 |
|
H5 |
C1 |
H6 |
108.579 |
| H7 |
C2 |
H8 |
107.153 |
|
H9 |
C3 |
H10 |
108.302 |
| H9 |
C3 |
H11 |
107.968 |
|
H10 |
C3 |
H11 |
107.970 |
Electronic energy levels
Charges, Dipole, Quadrupole and Polarizability
