|
Computational Chemistry Comparison and Benchmark DataBase
Release 22 (May 2022) Standard Reference Database 101
National Institute of Standards and Technology
|
|
|
|
You are here: Home > Geometry > Calculated > Calculated geometry OR Calculated > Geometry > Calculated geometry
|
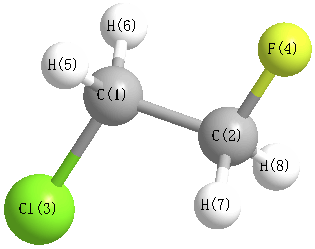
Geometry for (Ethane, 1-chloro-2-fluoro-)
1A' CS
1910171554
InChI=1S/C2H4ClF/c3-1-2-4/h1-2H2 INChIKey=VEZJSKSPVQQGIS-UHFFFAOYSA-N
MP2=FULL/aug-cc-pVTZ
Point group is Cs
| Atom |
Internal |
|
Principal |
| x (Å) |
y (Å) |
z (Å) |
|
a (Å) |
b (Å) |
c (Å) |
| C1 |
0.0000 |
0.6194 |
0.0000 |
|
-0.0216 |
0.6190 |
0.0000 |
| C2 |
0.9669 |
-0.5311 |
0.0000 |
|
0.9848 |
-0.4971 |
0.0000 |
| Cl3 |
-1.6486 |
-0.0323 |
0.0000 |
|
-1.6465 |
-0.0898 |
0.0000 |
| F4 |
2.2552 |
-0.0169 |
0.0000 |
|
2.2544 |
0.0617 |
0.0000 |
| H5 |
0.1240 |
1.2288 |
0.8866 |
|
0.0811 |
1.2324 |
0.8866 |
| H6 |
0.1240 |
1.2288 |
-0.8866 |
|
0.0811 |
1.2324 |
-0.8866 |
| H7 |
0.8404 |
-1.1430 |
0.8885 |
|
0.8798 |
-1.1130 |
0.8885 |
| H8 |
0.8404 |
-1.1430 |
-0.8885 |
|
0.8798 |
-1.1130 |
-0.8885 |
Atom - Atom Distances (Å)
| |
C1 |
C2 |
Cl3 |
F4 |
H5 |
H6 |
H7 |
H8 |
| C1 |
|
1.5028 |
1.7727 |
2.3432 |
1.0829 |
1.0829 |
2.1451 |
2.1451 |
|---|
| C2 |
1.5028 |
| 2.6626 |
1.3872 |
2.1433 |
2.1433 |
1.0862 |
1.0862 |
|---|
| Cl3 |
1.7727 |
2.6626 |
| 3.9038 |
2.3492 |
2.3492 |
2.8668 |
2.8668 |
|---|
| F4 |
2.3432 |
1.3872 |
3.9038 |
| 2.6229 |
2.6229 |
2.0147 |
2.0147 |
|---|
| H5 |
1.0829 |
2.1433 |
2.3492 |
2.6229 |
| 1.7731 |
2.4776 |
3.0478 |
|---|
| H6 |
1.0829 |
2.1433 |
2.3492 |
2.6229 |
1.7731 |
| 3.0478 |
2.4776 |
|---|
| H7 |
2.1451 |
1.0862 |
2.8668 |
2.0147 |
2.4776 |
3.0478 |
| 1.7770 |
|---|
| H8 |
2.1451 |
1.0862 |
2.8668 |
2.0147 |
3.0478 |
2.4776 |
1.7770 |
|
|---|
Maximum atom distance is 3.9038Å
between atoms Cl3 and F4.
Calculated Bond Angles (degrees) (Ignoring Hydrogens)
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
|
C1 |
C2 |
F4 |
108.284 |
|
C2 |
C1 |
Cl3 |
108.475 |
Calculated Bond Angles (degrees) (Involving Hydrogens)
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
|
C1 |
C2 |
H7 |
110.876 |
|
C1 |
C2 |
H8 |
110.876 |
|
C2 |
C1 |
H5 |
110.924 |
|
C2 |
C1 |
H6 |
110.924 |
|
Cl3 |
C1 |
H5 |
108.262 |
|
Cl3 |
C1 |
H6 |
108.262 |
|
F4 |
C2 |
H7 |
108.478 |
|
F4 |
C2 |
H8 |
108.478 |
|
H5 |
C1 |
H6 |
109.904 |
|
H7 |
C2 |
H8 |
109.773 |
For information on specific bond angles or dihedrals
see the geometry comparison page in section
Comparisons > Geometry > Bonds, angles.