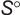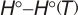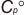.
| squib |
reference |
DOI |
| 1979HUB/HER |
Huber, K.P.; Herzberg, G., Molecular Spectra and Molecular Structure. IV. Constants of Diatomic Molecules, Van Nostrand Reinhold Co., 1979 |
10.1007/978-1-4757-0961-2 |
| 1992LBII/19c |
Landolt-Bornstein Numerical Data and Functional Relationships in Science and Technology, New Series, II/19c (1992) Spreinger-Verlag, Heidelberg |
|
| 1999Tam/Bro |
Filippo Tammassia, John M. Brown, Kenneth Evenson, J. Chem. Phys. 110, 7273, 1999 |
10.1063/1.478630 |
| 2000Tam/Bro:5523 |
Tamassia, F.; Brown, J.; Saita, S. "The detection of the free radical FO (X2 PI 3/2) by submillimeter-wave spectroscopy." Journal of Chemical Physics. 112, 5523-5526 (2000) |
10.1063/1.481184 |
| 2007Iri:389 |
KK Irikura "Experimental Vibrational Zero-Point Energies: Diatomic Molecules" J. Phys. Chem. Ref. Data 36(2), 389, 2007 |
10.1063/1.2436891 |
| Gurvich |
Gurvich, L.V.; Veyts, I. V.; Alcock, C. B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. Co., New York, 1989 |
|
| webbook |
NIST Chemistry Webbook (http://webbook.nist.gov/chemistry) |
10.18434/T4D303 |