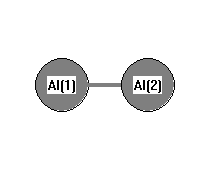
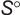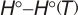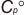.
| squib |
reference |
DOI |
| 1990Fu/Lem:8420 |
Z Fu, GW Lemire, GA Bishea, MD Morse "Spectroscopy and electronic structure fo jet-cooled Al2" J. Chem. Phys. 93(12), 8420, 1990 |
10.1063/1.459280 |
| 2002Rie/Tsc:231 |
JC Rienstra-Kiracofe, GS Tschumper, HF Schaefer III, S Nandi, GB Ellison "Atomic and Molecular Electron Affinities: Photoelectron Experiments and Theoretical Computations" Chemical Reviews 2002, 102, 231-282 |
10.1021/cr990044u |
| 2007Iri:389 |
KK Irikura "Experimental Vibrational Zero-Point Energies: Diatomic Molecules" J. Phys. Chem. Ref. Data 36(2), 389, 2007 |
10.1063/1.2436891 |
| Gurvich |
Gurvich, L.V.; Veyts, I. V.; Alcock, C. B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. Co., New York, 1989 |
|
| NSRDS-NBS10 |
R. D. Nelson Jr., D. R. Lide, A. A. Maryott "Selected Values of electric dipole moments for molecules in the gas phase" NSRDS-NBS10, 1967 |
10.6028/NBS.NSRDS.10 |
| webbook |
NIST Chemistry Webbook (http://webbook.nist.gov/chemistry) |
10.18434/T4D303 |