.
| squib |
reference |
DOI |
| 1976Hellwege(II/7) |
Hellwege, KH and AM Hellwege (ed.). Landolt-Bornstein: Group II: Atomic and Molecular Physics Volume 7: Structure Data of Free Polyatomic Molecules. Springer-Verlag. Berlin. 1976. |
|
| 2000Bal/Bal:517 |
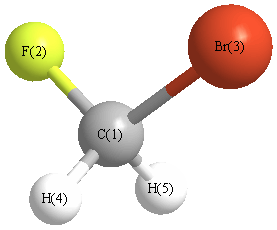
A Baldacci, A Baldan, A Gambi, P Stoppa "The vibrational spectra and normal coordiantes analysis of bromofluoromethane, CH2BrF" J. Mol. Struct. 517-518 (2000) 197-208 |
10.1016/S0022-2860(99)00251-3 |
| 2007Bal/Sto:126 |
A Baldacci, P Stoppa, A Pietropolli Charmet, S Giorgianni, G Cazzoli, L Cludi, C Puzzarini, R Wugt Larsen, "Spectroscopic constants of the ground and lower vibrational states of CH2 81BrF: A combined high resolution infrared and microwave study" J. Mol. Spec. 246 (2007) 126-132 |
10.1016/j.jms.2007.09.002 |
| 2007Caz/Puz:112 |
G Cazzoli, C Puzzarini, A Baldacci, A Baldan "Determination of the molecular dipole moment of bromofluoromethane: Microwave Stark spectra and ab initio calculations" J. Mol. Spec. 241 (2007) 112-115 |
10.1016/j.jms.2006.11.004 |
| TRC |
Frenkel, M; Marsh, K.N.; Wilhoit, R.C.; Kabo, G.J.; Roganov, G.N.,Thermodynamics of Organic Compounds in the Gas State,Thermodynamics Research Center, College Station, TX, 1994 |
|