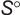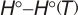Geometric Data
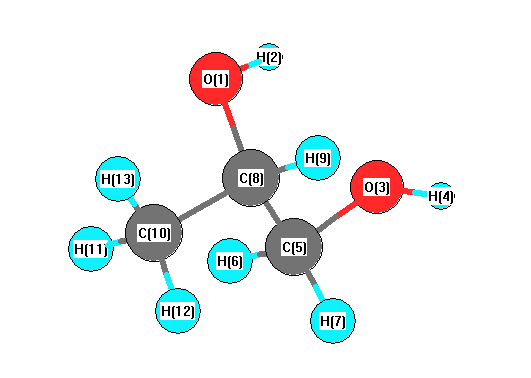
Point Group C1
Internal coordinates
distances (r) in Å, angles (a) in degrees, dihedrals (d) in degrees
These cartesians were determined using some assumed coordinate values.
Cartesians
| Atom |
x (Å) |
y (Å) |
z (Å) |
| O1 |
0.7246 |
1.2702 |
-0.4174 |
| H2 |
0.1040 |
2.0031 |
-0.1384 |
| O3 |
-1.5556 |
-0.2721 |
-0.5724 |
| H4 |
-0.9884 |
-0.9396 |
-1.0547 |
| C5 |
-0.9608 |
-0.0235 |
0.6929 |
| H6 |
-1.4120 |
0.9122 |
1.0392 |
| H7 |
-1.1267 |
-0.9398 |
1.2690 |
| C8 |
0.5503 |
0.1827 |
0.4789 |
| H9 |
1.0258 |
0.4207 |
1.4361 |
| C10 |
1.2254 |
-1.0572 |
-0.1362 |
| H11 |
0.7714 |
-1.2761 |
-1.1084 |
| H12 |
2.2948 |
-0.8631 |
-0.2688 |
| H13 |
1.0906 |
-1.9153 |
0.5305 |
Atom - Atom Distances 
Distances in Å
| |
O1 |
H2 |
O3 |
H4 |
C5 |
H6 |
H7 |
C8 |
H9 |
C10 |
H11 |
H12 |
H13 |
| O1 |
|
1.0000 | 2.7572 | 2.8677 | 2.3972 | 2.6105 | 3.3399 | 1.4200 | 2.0610 | 2.3972 | 2.6388 | 2.6531 | 3.3437 |
| H2 |
1.0000 |
|
2.8494 | 3.2699 | 2.4355 | 2.2079 | 3.4865 | 1.9733 | 2.4151 | 3.2593 | 3.4841 | 3.6099 | 4.0957 |
| O3 |
2.7572 | 2.8494 |
|
1.0000 | 1.4200 | 2.0051 | 2.0051 | 2.3972 | 3.3433 | 2.9224 | 2.5904 | 3.9074 | 3.3045 |
| H4 |
2.8677 | 3.2699 | 1.0000 |
|
1.9733 | 2.8272 | 2.3278 | 2.4452 | 3.4802 | 2.3996 | 1.7925 | 3.3768 | 2.7906 |
| C5 |
2.3972 | 2.4355 | 1.4200 | 1.9733 |
|
1.0950 | 1.0950 | 1.5400 | 2.1671 | 2.5564 | 2.7953 | 3.4970 | 2.7953 |
| H6 |
2.6105 | 2.2079 | 2.0051 | 2.8272 | 1.0950 |
|
1.8879 | 2.1671 | 2.5183 | 3.4951 | 3.7640 | 4.3131 | 3.8101 |
| H7 |
3.3399 | 3.4865 | 2.0051 | 2.3278 | 1.0950 | 1.8879 |
|
2.1671 | 2.5519 | 2.7424 | 3.0607 | 3.7521 | 2.5326 |
| C8 |
1.4200 | 1.9733 | 2.3972 | 2.4452 | 1.5400 | 2.1671 | 2.1671 |
|
1.0950 | 1.5400 | 2.1671 | 2.1671 | 2.1671 |
| H9 |
2.0610 | 2.4151 | 3.3433 | 3.4802 | 2.1671 | 2.5183 | 2.5519 | 1.0950 |
|
2.1671 | 3.0689 | 2.4830 | 2.5063 |
| C10 |
2.3972 | 3.2593 | 2.9224 | 2.3996 | 2.5564 | 3.4951 | 2.7424 | 1.5400 | 2.1671 |
|
1.0950 | 1.0950 | 1.0950 |
| H11 |
2.6388 | 3.4841 | 2.5904 | 1.7925 | 2.7953 | 3.7640 | 3.0607 | 2.1671 | 3.0689 | 1.0950 |
|
1.7878 | 1.7879 |
| H12 |
2.6531 | 3.6099 | 3.9074 | 3.3768 | 3.4970 | 4.3131 | 3.7521 | 2.1671 | 2.4830 | 1.0950 | 1.7878 |
|
1.7878 |
| H13 |
3.3437 | 4.0957 | 3.3045 | 2.7906 | 2.7953 | 3.8101 | 2.5326 | 2.1671 | 2.5063 | 1.0950 | 1.7879 | 1.7878 |
|
Calculated geometries
for C
3H
8O
2 (Propylene glycol).
Experimental Bond Angles (degrees) from cartesians 
| atom1 |
atom2 |
atom3 |
angle |
|
atom1 |
atom2 |
atom3 |
angle |
| O1 |
C8 |
C5 |
108.100 |
|
O1 |
C8 |
H9 |
109.388 |
| O1 |
C8 |
C10 |
108.100 |
|
H2 |
O1 |
C8 |
108.000 |
| O3 |
C5 |
H6 |
105.000 |
|
O3 |
C5 |
H7 |
105.000 |
| O3 |
C5 |
C8 |
108.100 |
|
H4 |
O3 |
C5 |
108.000 |
| C5 |
C8 |
H9 |
109.500 |
|
C5 |
C8 |
C10 |
112.200 |
| H6 |
C5 |
H7 |
119.092 |
|
H6 |
C5 |
C8 |
109.500 |
| H7 |
C5 |
C8 |
109.500 |
|
C8 |
C10 |
H11 |
109.500 |
| C8 |
C10 |
H12 |
109.500 |
|
C8 |
C10 |
H13 |
109.500 |
| H9 |
C8 |
C10 |
109.500 |
|
H11 |
C10 |
H12 |
109.440 |
| H11 |
C10 |
H13 |
109.447 |
|
H12 |
C10 |
H13 |
109.440 |
Bond descriptions
Examples: C-C single bond, C=C, double bond, C#C triple bond, C:C aromatic bond
| Bond Type |
Count |
| H-C |
6 |
| H-O |
2 |
| C-C |
2 |
| C-O |
2 |
Connectivity
| Atom 1 |
Atom 2 |
| O1 |
H2 |
| O1 |
C8 |
| O3 |
H4 |
| O3 |
C5 |
| C5 |
H6 |
| C5 |
H7 |
| C5 |
C8 |
| C8 |
H9 |
| C8 |
C10 |
| C10 |
H11 |
| C10 |
H12 |
| C10 |
H13 |