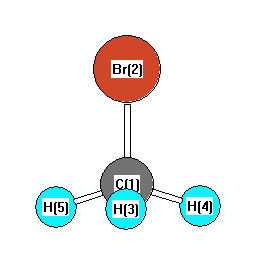
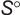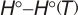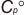.
| squib |
reference |
DOI |
| 1974Hel/Hel(II/6) |
Hellwege, KH and AM Hellwege (eds.). Landolt-Bornstein: Group II: Volume 6 Molecular Constants from Microwave, Molecular Beam, and Electron Spin Resonance Spectroscopy Springer-Verlag. Berlin. 1974. |
10.1007/b19951 |
| 1981Gra:394 |
G Graner " The methyl bromide molecule: A critical consideration of perturbations in spectra" J. Mol. Spec. 90, 394-438, (1981) |
10.1016/0022-2852(81)90136-3 |
| 1984Gra/Gub |
CG Gray, KE Gubbins "Theory of molecular fluids. Volume 1:Fundamentals" Clarendon Press, Oxford 1984 |
|
| 1998Kuc |
K Kuchitsu(ed) "Structure of Free Polyatomic Molecules - Basic Data" Springer, Berlin, 1998 |
10.1007/978-3-642-45748-7 |
| Gurvich |
Gurvich, L.V.; Veyts, I. V.; Alcock, C. B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. Co., New York, 1989 |
|
| Shim |
Shimanouchi, T. , Tables of Molecular Vibrational Frequencies, Consolidated Volu |
10.6028/NBS.NSRDS.39 |