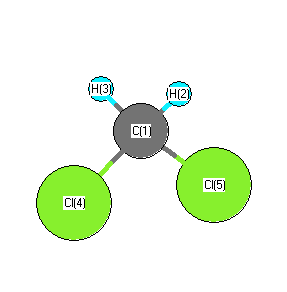
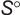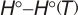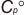.
| squib |
reference |
DOI |
| 1952Mye/Gwi:1420-1427 |
RJ Myers, WD Gwinn "The Microwave Spectra, Structure, Dipole Moment, and Chlorine Nuclear Quadrupole Coupling Constants of Methylene Chloride" J. Chem. Phys. 20, 1420 (1952) |
10.1063/1.1700773 |
| 1989Tul/Niv:355 |
Tullini, F.; Nivellini, G.D.; Carlotti, M. "The Far-Infrared Spectrum of Methylene Chloride." Journal of Molecular Spectroscopy. 138, 355-374 (1989) |
10.1016/0022-2852(89)90004-0 |
| 1998Gus/Rui:163 |
M Gussoni, R Rui, G Zerbi "Electronic and relaxation contribution to linear molecular polarizability. An analysis of the experimental values" J. Mol. Struct. 447 (1998) 163-215 |
10.1016/S0022-2860(97)00292-5 |
| 1998Kuc |
K Kuchitsu(ed) "Structure of Free Polyatomic Molecules - Basic Data" Springer, Berlin, 1998 |
10.1007/978-3-642-45748-7 |
| Gurvich |
Gurvich, L.V.; Veyts, I. V.; Alcock, C. B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. Co., New York, 1989 |
|
| Shim |
Shimanouchi, T. , Tables of Molecular Vibrational Frequencies, Consolidated Volu |
10.6028/NBS.NSRDS.39 |
| webbook |
NIST Chemistry Webbook (http://webbook.nist.gov/chemistry) |
10.18434/T4D303 |