.
| squib |
reference |
DOI |
| 1974sve/kov |
L.M. Sverdlov, M. A. Kovner, E. P. Krainov, "Vibrational Spectra of Polyatomic Molecules" Wiley, New York 1974 |
|
| 1978hir:145 |
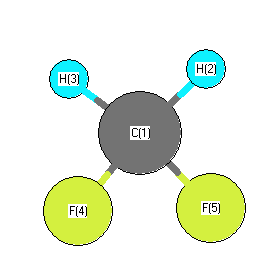
Hirota, H. Anharmonic Potential Function and Equilibrium Structure of Methylene Fluoride, J. Mol. Spec. 71, 145-159, 1978 |
10.1016/0022-2852(78)90079-6 |
| 1978Hir:409 |
Hirota, E. "Microwave Spectrum of Methylene Fluoride in Excited Vibrational States." Journal of Molecular Spectroscopy. 69, 409-420 (1978) |
10.1016/0022-2852(78)90234-5 |
| 1980Kon/Nak:5409 |
S Kondo, T Nakanaga, S Saeki,"Infrared intensities and Coriolis interactions in methylene fluoride" J. Chem. Phys. 73(11), 5409, 1980 |
10.1063/1.440086 |
| 1984Gra/Gub |
CG Gray, KE Gubbins "Theory of molecular fluids. Volume 1:Fundamentals" Clarendon Press, Oxford 1984 |
|
| 1998Gus/Rui:163 |
M Gussoni, R Rui, G Zerbi "Electronic and relaxation contribution to linear molecular polarizability. An analysis of the experimental values" J. Mol. Struct. 447 (1998) 163-215 |
10.1016/S0022-2860(97)00292-5 |
| Gurvich |
Gurvich, L.V.; Veyts, I. V.; Alcock, C. B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. Co., New York, 1989 |
|
| NSRDS-NBS10 |
R. D. Nelson Jr., D. R. Lide, A. A. Maryott "Selected Values of electric dipole moments for molecules in the gas phase" NSRDS-NBS10, 1967 |
10.6028/NBS.NSRDS.10 |
| webbook |
NIST Chemistry Webbook (http://webbook.nist.gov/chemistry) |
10.18434/T4D303 |