.
| squib |
reference |
DOI |
| 1971Fly/Ben:225 |
WH Flygare, RC Benson "The molecular Zeeman effect in diamagnetic molecules and the determination of molecular magnetic moments (g values), magnetic susceptibilities, and molecular quadrupole moments" Mol. Phys. 1971, 20 (2), 225-250 |
10.1080/00268977100100221 |
| 1974Hir/Sug:251 |
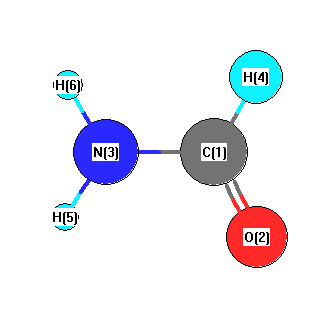
E Hirota, R Sugisaki "Molecular Structure and Internal Motion of Formamide from Microwave Spectrum" J. Mol. Spec. 49, 251, 1974 |
10.1016/0022-2852(74)90274-4 |
| 1983Sug/Ham:1045 |
Y Sugawara, Y Hamada, M Tsuboi, " Vibration-Rotation Spectra of Formamides" Bull. Chem. Soc. Jpn. 56, 1045, 1983 |
10.1246/bcsj.56.1045 |
| 1998Gus/Rui:163 |
M Gussoni, R Rui, G Zerbi "Electronic and relaxation contribution to linear molecular polarizability. An analysis of the experimental values" J. Mol. Struct. 447 (1998) 163-215 |
10.1016/S0022-2860(97)00292-5 |
| NSRDS-NBS10 |
R. D. Nelson Jr., D. R. Lide, A. A. Maryott "Selected Values of electric dipole moments for molecules in the gas phase" NSRDS-NBS10, 1967 |
10.6028/NBS.NSRDS.10 |
| webbook |
NIST Chemistry Webbook (http://webbook.nist.gov/chemistry) |
10.18434/T4D303 |