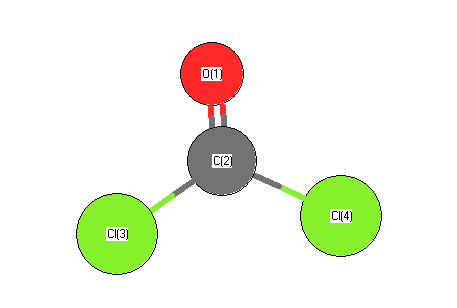
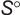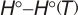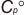.
| squib |
reference |
DOI |
| 1966Herzberg |
Herzberg, G., Electronic spectra and electronic structure of polyatomic molecules,Van Nostrand,New York, 1966 |
|
| 1968Hop/Rus:3765 |
MJ Hopper, JW Russell, J Overend "Vibrational Intensities. XVI. COF2, COCL2, COBr2" J. Chem. Phys. 48(8), 3765, 1968 |
10.1063/1.1669683 |
| 1998Gus/Rui:163 |
M Gussoni, R Rui, G Zerbi "Electronic and relaxation contribution to linear molecular polarizability. An analysis of the experimental values" J. Mol. Struct. 447 (1998) 163-215 |
10.1016/S0022-2860(97)00292-5 |
| 1998Kuc |
K Kuchitsu(ed) "Structure of Free Polyatomic Molecules - Basic Data" Springer, Berlin, 1998 |
10.1007/978-3-642-45748-7 |
| JANAF |
Chase, M.W., Jr.; Davies, C.A.; Downey, J.R., Jr.; Frurip, D.J.; McDonald, R.A.; Syverud, A.N., JANAF Thermochemical Tables (Third Edition), J. Phys. Chem. Ref. Data,Suppl. 1, 1985, 14, 1. |
|
| NSRDS-NBS10 |
R. D. Nelson Jr., D. R. Lide, A. A. Maryott "Selected Values of electric dipole moments for molecules in the gas phase" NSRDS-NBS10, 1967 |
10.6028/NBS.NSRDS.10 |
| webbook |
NIST Chemistry Webbook (http://webbook.nist.gov/chemistry) |
10.18434/T4D303 |