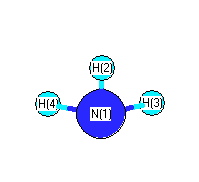
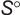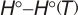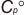.
| squib |
reference |
DOI |
| 1966Herzberg |
Herzberg, G., Electronic spectra and electronic structure of polyatomic molecules,Van Nostrand,New York, 1966 |
|
| 1970Shi:3572-3576 |
F Shimizu "Stark Spectroscopy of NH3 ν2 Band by 10-μ CO2 and N2O Lasers" J. Chem. Phys. 52, 3572 (1970) |
10.1063/1.1673524 |
| 1974Hel/Hel(II/6) |
Hellwege, KH and AM Hellwege (eds.). Landolt-Bornstein: Group II: Volume 6 Molecular Constants from Microwave, Molecular Beam, and Electron Spin Resonance Spectroscopy Springer-Verlag. Berlin. 1974. |
10.1007/b19951 |
| 1983Koo/Vis:203 |
Th Koops, T Visser, WMA Smit "Measurement and Interpretation of the Absolute Infrared Intensities of NH3 and ND3" Journal of Molecular Structure 96 (1983) 203-218 |
10.1016/0022-2860(83)90049-2 |
| 1997Oln/Can:59 |
TN Olney, NM Cann, G Cooper, CE Brion, Absolute scale determination for photoabsorption spectra and the calculation of molecular properties using dipole sum-rules, Chem. Phys. 223 (1997) 59-98 |
10.1016/S0301-0104(97)00145-6 |
| CODATA |
Cox, J.D.; Wagman, D.D.; Medvedev, V.A.CODATA Key Values for Thermodynamics. Hemisphere, New York, 1989 |
10.1002/bbpc.19900940121 |
| Gurvich |
Gurvich, L.V.; Veyts, I. V.; Alcock, C. B., Thermodynamic Properties of Individual Substances, Fouth Edition, Hemisphere Pub. Co., New York, 1989 |
|
| Shim |
Shimanouchi, T. , Tables of Molecular Vibrational Frequencies, Consolidated Volu |
10.6028/NBS.NSRDS.39 |
| webbook |
NIST Chemistry Webbook (http://webbook.nist.gov/chemistry) |
10.18434/T4D303 |